Description
Comprehensive Clinical Analysis of Osteomyelitis: A Multidisciplinary Guide to Pathogenesis, Diagnostics, and Integrative Management
The healing of human bone, a structure of both immense strength and intricate biological vitality, remains one of the most profound expressions of the body’s innate drive toward equilibrium. Osteomyelitis, defined as an inflammatory process of the bone and bone marrow resulting from microbial infection, represents a significant disruption of this balance. Known to the ancients and feared throughout the history of medicine, this condition transforms a typically sterile and mineralized environment into a site of progressive destruction, necrosis, and potentially life-altering morbidity. In the contemporary clinical landscape, the management of osteomyelitis is not merely an exercise in antimicrobial selection but a sophisticated orchestration of diagnostic precision, surgical skill, and an empathetic understanding of the patient’s physiological and psychological landscape. This report serves as a bridge between the rigorous standards of internal medicine, established by the zolden standard of Harrison’s Principles, and the compassionate, holistic care required to restore the patient to wholeness.
Definition and Etymological Origins
Osteomyelitis derives its name from the Greek roots osteon (bone), myelo (marrow), and itis (inflammation). While the term technically encompasses any inflammation of the bone, in the clinical setting, it almost exclusively refers to an infectious etiology, most commonly bacterial. It is characterized by the presence of microorganisms that induce local bone destruction, vascular compromise, and the subsequent formation of necrotic bone segments. The pathology is characterized by its chronicity and its ability to persist despite the body’s immune responses, often requiring a prolonged and multifaceted therapeutic approach to achieve true resolution.
The nature of osteomyelitis is profoundly influenced by the route of infection, the virulence of the invading pathogen, and the underlying health of the host. Whether through the silent seeding of the bloodstream in a child or the direct inoculation following an orthopedic repair in an adult, the disease process represents a failure of the body’s barriers and a challenge to the principles of “Primum non nocere,” as clinicians must balance aggressive treatment with the preservation of functional limb and life.
Epidemiology and Demographic Variations
The epidemiology of osteomyelitis is a reflection of the evolving risks in modern society, where advances in surgical intervention and the survival of immunocompromised individuals have created new niches for bone infection. While historically a disease of childhood trauma, the burden has shifted toward aging populations with vascular comorbidities and those undergoing complex reconstructive surgeries.
Global Incidence and Predilection
In the pediatric population, acute hematogenous osteomyelitis remains the most common form, with an estimated incidence of 20 per 100,000 children annually. In this demographic, males are affected more frequently than females, likely due to a higher frequency of blunt trauma, which can create micro-hematomas in the bone that act as a nidus for bacteremic seeding. In adults, the incidence is increasingly associated with the surgical placement of hardware and the growing epidemic of diabetes mellitus.
The risk of infection following joint replacement (arthroplasty) ranges from 0.3% to 2.4% for the hip and 1.0% to 3.0% for the knee. While these percentages may seem small, the absolute number of cases is significant given the volume of such procedures globally. Fracture-associated infections are even more prevalent, particularly in open fractures where the skin barrier is catastrophically breached. High-energy open fractures of the lower extremities carry an infection risk as high as 27%, with the tibia being the most frequently involved bone due to its limited soft tissue coverage.
Demographic Trends
The distribution of osteomyelitis is also influenced by socioeconomic and environmental factors. Individuals from lower socioeconomic backgrounds are more prone to presenting with chronic forms of the disease due to delays in initial diagnosis or incomplete treatment of acute episodes. Geographically, certain types of osteomyelitis, such as those caused by Burkholderia pseudomallei or specific fungal pathogens, show regional clusters in tropical or arid environments, respectively.
| Demographic Factor | Predominant Patterns and Associations |
| Age Group: Children | Primarily hematogenous spread; involves long bone metaphyses; S. aureus is the lead pathogen. |
| Age Group: Elderly | Primarily vertebral involvement; associated with chronic urinary or skin infections. |
| Gender | Consistently higher incidence in males across all age categories. |
| Diabetes | Strong association with lower extremity (foot) osteomyelitis; often polymicrobial. |
| Iatrogenic | Post-surgical infections; often involves biofilm-forming Staphylococci on hardware. |
Classification of the Pathology
A nuanced understanding of osteomyelitis requires a classification system that accounts for the duration of the infection, the mechanism of spread, and the physiological status of the patient. While no single system is universally accepted, the Waldvogel and Cierny-Mader systems provide the most comprehensive frameworks for clinical practice.
The Waldvogel Classification: Etiology and Duration
The Waldvogel system, introduced in 1970, categorizes osteomyelitis based on its origin and clinical course. This system is particularly useful for the initial diagnostic workup and understanding the likely pathogens involved.
| Category | Pathogenesis and Clinical Features |
| Hematogenous | Originates from a bacteremic episode; common in children (long bones) and adults (vertebrae). |
| Contiguous Focus | Spreads from an adjacent infection, such as cellulitis, or direct inoculation from trauma/surgery. |
| Vascular Insufficiency | Associated with diabetes and peripheral arterial disease; primarily involves the small bones of the feet. |
| Duration: Acute | Presents within days to weeks; characterized by rapid onset and systemic toxemia. |
| Duration: Chronic | Persists for months or years; characterized by necrotic bone (sequestra) and sinus tracts. |
The Cierny-Mader Staging System: Anatomy and Host Physiology
For surgical planning and prognostic assessment, the Cierny-Mader system is the gold standard. It recognizes that the success of treatment depends as much on the patient’s internal healing environment as it does on the anatomical extent of the infection.
Anatomical Types:
- Stage 1: Medullary Osteomyelitis – The infection is confined to the marrow space; often seen in early hematogenous spread or after intramedullary rod placement.
- Stage 2: Superficial Osteomyelitis – The infection is limited to the outer cortex, typically occurring from contiguous soft tissue defects or chronic ulcers.
- Stage 3: Localized Osteomyelitis – A well-defined sequestrum involves the full thickness of the cortex, but the bone remains stable.
- Stage 4: Diffuse Osteomyelitis – The infection permeates the entire bone segment, resulting in loss of mechanical stability before or after surgical debridement.
Physiological Host Classes:
- Class A Host: A patient with a healthy immune system, normal metabolic function, and excellent vascularity.
- Class B Host: A patient with local compromise (Bl), such as venous stasis or scarring, or systemic compromise (Bs), such as malnutrition or diabetes.
- Class C Host: A patient for whom the treatment’s morbidity (e.g., radical surgery or high-risk antibiotics) outweighs the benefits of curing the infection.
Pathophysiology of Bone Infection
The pathophysiology of osteomyelitis is an intricate battle between microbial evasion strategies and the host’s inflammatory response, set against the rigid and unique architecture of the skeletal system. Following the principles of Harrison’s Internal Medicine, the hallmarks of this process are vascular compromise and tissue necrosis.
Mechanism of Infection and Spread
Pathogens reach the bone through three primary routes: hematogenous seeding, contiguous spread, and direct inoculation. In hematogenous osteomyelitis, the specific location of the infection is determined by age-related vascular patterns. In children, the infection usually begins in the metaphysis of long bones, where the sluggish flow through large venous sinusoids and the presence of terminal capillary loops facilitate bacterial adherence. In adults, these metaphyseal vessels disappear, and the vertebral column becomes the primary target due to its persistent and robust blood supply.
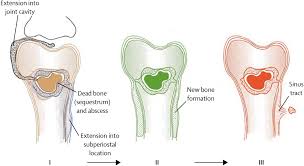
The Microenvironment: Sequestrum and Involucrum
Once bacteria establish a foothold, they induce an acute inflammatory response. The resulting increase in intramedullary pressure leads to vascular stasis and thrombosis of small vessels. This ischemia results in the death of bone tissue, creating a sequestrum—a piece of dead, infected bone that has become detached from its blood supply. Because it is avascular, the sequestrum acts as a sanctuary for bacteria, shielding them from systemic antibiotics and the host’s immune cells.
The lifting of the periosteum by subperiosteal abscesses further impairs the blood supply to the cortex. In a remarkable attempt at repair, the periosteum begins to lay down a layer of reactive, viable new bone around the site of infection. This new bone is known as the involucrum. If the pus breaks through the involucrum to reach the skin, it forms a cloaca—the opening of a sinus tract through which necrotic debris and exudate are discharged.
Biofilms and Persister Cells
A critical aspect of the pathophysiology is the formation of biofilms, particularly by Staphylococcus aureus and Staphylococcus epidermidis. Biofilms are structured communities of bacteria encased in a self-produced matrix of extracellular polymeric substances. Within this matrix, bacteria can enter a state of low metabolic activity (persister cells), making them highly resistant to standard bactericidal concentrations of antibiotics. This phenomenon explains why hardware-related infections and chronic osteomyelitis are so difficult to eradicate without the complete surgical removal of all foreign material and necrotic tissue.
Microbiology
The microbial spectrum of osteomyelitis is diverse, though certain pathogens dominate specific clinical scenarios.
- Staphylococcus aureus: The leading cause of both acute and chronic osteomyelitis in all age groups, responsible for approximately 60% of hematogenous cases.
- Coagulase-negative Staphylococci: Often associated with prosthetic joints and surgical hardware.
- Pseudomonas aeruginosa and Serratia marcescens: Frequently isolated in injection drug users and in the context of puncture wounds.
- Salmonella species: Highly prevalent in patients with sickle cell disease, where micro-infarctions of the gut mucosa allow for bacteremic spread to infarcted marrow.
- Mycobacterium tuberculosis: Known as Pott’s disease when involving the spine, it typically results in significant bony destruction and kyphotic deformity.
Clinical Manifestation of the Pathology
The clinical presentation of osteomyelitis varies according to the site of infection and the patient’s age, ranging from the dramatic presentation of systemic sepsis to the silent progression of a diabetic foot ulcer.
Pediatric Acute Hematogenous Osteomyelitis
Children often present with a rapid onset of high fever, malaise, and localized signs of inflammation. A critical sign is “pseudoparalysis,” where the child refuses to use the affected limb or cries when it is moved. The pain is typically described as deep and boring, maximal over the involved metaphysis. Approximately 50% of these patients may present with more vague symptoms, requiring a high degree of clinical suspicion.
Adult Vertebral Osteomyelitis
The presentation in adults is usually more insidious, with localized back pain developing over 1 to 3 months. Fever is present in only about 50% of cases, and systemic symptoms are often mild. The pain is typically not relieved by rest and may be exacerbated by percussion over the involved vertebrae. Neurologic symptoms, such as weakness or sensory deficits, occur in approximately 10% of cases and signal the presence of an epidural abscess or spinal instability.
Contiguous and Chronic Presentations
Osteomyelitis following surgery or trauma often presents as a wound that is slow to heal or as persistent drainage through a sinus tract. In chronic osteomyelitis, systemic symptoms are frequently absent, and the primary complaint may be a longstanding, intermittently discharging ulcer or deep-seated, dull pain.
Vascular Insufficiency and Diabetes
In patients with diabetes, osteomyelitis is often discovered during the evaluation of a chronic foot ulcer. Because of peripheral neuropathy, the patient may not feel pain, and the infection can progress to involve the small bones of the feet before being clinically detected. Redness, heat, and swelling of the foot (the “hot foot”) in a diabetic patient should always raise suspicion of underlying osteomyelitis or Charcot neuroarthropathy.
Diagnostics of the Pathology
The diagnostic pathway for osteomyelitis is a stepwise integration of clinical examination, laboratory tests, and specialized imaging, always aiming toward the gold standard of tissue diagnosis.
Physical Examination Findings
The physical examination should focus on identifying signs of localized inflammation and functional impairment. Key maneuvers include:
- Palpation for Point Tenderness: Especially important in pediatric cases and vertebral osteomyelitis.
- The “Probe-to-Bone” Test: In diabetic foot ulcers, the ability to reach bone with a sterile metal probe has high predictive value for osteomyelitis.
- Assessment of Range of Motion: Joint effusions may be seen if the infection is near a joint, though the joint itself may remain sterile (sympathetic effusion).
- Inspection for Sinus Tracts: A persistent sinus tract is pathognomonic for chronic osteomyelitis.
Laboratory Analysis
Laboratory tests are useful for supporting the diagnosis and monitoring the response to therapy, although they lack specificity.
- ESR and CRP: Both are typically elevated in acute osteomyelitis. CRP is the preferred marker for monitoring as it rises and falls more rapidly in response to effective treatment. An ESR > 70 mm/hr in a diabetic patient with a foot ulcer is highly suggestive of underlying bone involvement.
- White Blood Cell (WBC) Count: Leukocytosis is common in acute cases but is frequently normal in chronic osteomyelitis.
- Procalcitonin: A novel marker that can help predict the severity of the infective process.
- Blood Cultures: Positive in approximately 40% to 50% of pediatric hematogenous cases and adult vertebral cases, providing a non-invasive means of identifying the pathogen.
Imaging Studies
Imaging is essential for determining the anatomical extent of the infection and guiding surgical or biopsy procedures.
- Plain Radiography (X-ray): The initial test. Early changes include soft tissue swelling and periosteal elevation. Bony destruction (lysis) and sclerosis typically lag behind the clinical infection by 10 to 14 days.
- Magnetic Resonance Imaging (MRI): The modality of choice for early diagnosis. It can detect bone marrow edema within 3 to 5 days of symptom onset. It is highly sensitive (97%) for vertebral osteomyelitis and superior for identifying epidural abscesses.
- Computed Tomography (CT): More sensitive than MRI for identifying sequestra, involucrum, and gas within the bone. It is also the preferred guide for percutaneous biopsy.
- PET-CT (18F-FDG): An increasingly valuable tool with high sensitivity and specificity (>90%). It is particularly useful in differentiating chronic osteomyelitis from post-surgical changes and for detecting metastatic foci of infection in bacteremic patients.
- Bone Scintigraphy (Bone Scan): Uses Technetium-99m to identify areas of increased bone turnover. It is highly sensitive within 48 hours but lacks specificity, as it cannot easily distinguish between infection, fracture, or tumor.
| Imaging Modality | Sensitivity | Specificity | Primary Clinical Use |
| X-ray | 43% – 75% | 75% – 83% | Initial screening; detecting chronic changes. |
| MRI | 90% – 100% | 70% – 93% | Early diagnosis; spinal and soft tissue evaluation. |
| CT | Moderate | High (for bone) | Identifying sequestra; guiding biopsies. |
| FDG PET/CT | >95% | 75% – 99% | Chronic disease; metastatic infection screening. |
| Bone Scan | High | Low | Screening for multiple sites; vertebral cases. |
The Gold Standard: Bone Biopsy
A definitive diagnosis of osteomyelitis requires the identification of the pathogen and characteristic histopathological findings (e.g., bone necrosis, inflammatory cells) from a tissue specimen. Percutaneous needle aspiration or open biopsy should be performed before starting antibiotics, except in patients with clinical instability or positive blood cultures that clearly explain the clinical picture.
Differential Diagnosis (French’s Index Approach)
Differentiating osteomyelitis from conditions with similar presentations requires an analytical approach that balances probability with the severity of potential misdiagnosis. Following the structure of French’s Index, symptoms of bone pain and swelling are categorized to ensure no “red flags” are overlooked.
Localized Bone Pain and Swelling
| Condition | Probability | Severity | Distinguishing Clinical and Imaging Features |
| Acute Fracture | High (with trauma) | Moderate | History of injury; sharp pain; systemic symptoms absent; X-ray shows fracture line. |
| Acute Gout | High (adults) | Low | Primarily joint-based; history of previous attacks; presence of urate crystals in joint aspirate. |
| Cellulitis | High | Low | Soft tissue inflammation only; bone marrow signal on MRI is normal; no bony destruction on X-ray. |
| Septic Arthritis | Moderate | High | Sudden joint immobility; high WBC in joint fluid; osteomyelitis can coexist, especially in the hip. |
| Ewing’s Sarcoma | Low | Very High | Seen in children/adolescents; systemic symptoms possible; X-ray shows “onion-skin” periosteal reaction. |
| Bone Infarction | Moderate (Sickle Cell) | Moderate | Difficulty differentiating from osteomyelitis; often follows a “crisis” event; afebrile; MRI findings may overlap. |
| Charcot Joint | Moderate (Diabetes) | Moderate | Relatively painless despite severe deformity; associated with neuropathy; chronic course; limited systemic signs. |
| Gaucher’s Disease | Very Low | Moderate | “Bone crises” in patients with splenomegaly and bruising; genetic predisposition (Ashkenazi Jews). |
Therapeutic Strategies and Pharmacotherapy
The treatment of osteomyelitis is a journey of endurance, requiring high-dose, targeted antimicrobial therapy and, in many cases, definitive surgical intervention to restore the bone’s vitality.
Pharmacotherapeutic Goals
The primary goals of therapy are to eradicate the infecting pathogen, prevent the progression to chronic disease or the emergence of resistance, and restore the patient’s functional status. This requires achieving therapeutic concentrations of antibiotics within the poorly vascularized and rigid bone matrix.
Non-pharmacological and Holistic Therapies
Integrative medicine focuses on optimizing the host’s healing environment to support the effectiveness of conventional treatments.
- Optimal Healing Environment (OHE): Fostering a relationship of trust and using stress-reduction techniques to lower cortisol levels, which can otherwise suppress immune function.
- Anti-inflammatory Diet: A diet rich in phytonutrients and low in processed sugars may help modulate systemic inflammation.
- Hyperbaric Oxygen Therapy (HBOT): Indicated for refractory chronic osteomyelitis. By increasing oxygen tension in hypoxic bone, HBOT enhances the bactericidal capacity of neutrophils and promotes angiogenesis.
- Nutritional Supplements: Support for the immune system may include Vitamin C (500mg twice daily), Zinc (30-50mg daily), and Vitamin E. Probiotics (Lactobacillus/Bifidobacteria) are essential to maintain gut health during prolonged antibiotic therapy.
- Acupuncture: May be used as a supportive therapy to manage chronic pain and reduce inflammation, though it must not be a substitute for antibiotics.
Classical Pharmacotherapy: Strategies and Dosages
Antibiotic therapy must be guided by culture and susceptibility results whenever possible. The choice of agent depends on the pathogen, the presence of foreign material, and the patient’s renal and hepatic function.
| Pathogen / Context | First-Choice Antibiotic (IV) | Adult Dosage Information | Oral Step-Down Options |
| MSSA | Cefazolin or Nafcillin | Cefazolin: 2g q8h; Nafcillin: 2g q4h. | Cephalexin (1g q6h) or Dicloxacillin. |
| MRSA | Vancomycin | 15-20 mg/kg q8-12h (Target trough 15-20 $\mu g/mL$). | Linezolid (600mg BID) or TMP-SMX. |
| Pseudomonas | Cefepime or Piperacillin-Tazobactam | Cefepime: 2g q8-12h; Pip-Tazo: 4.5g q6-8h. | Ciprofloxacin (750mg BID). |
| Staph (Vertebral) | Vancomycin | 15-20 mg/kg q12h. | Levofloxacin + Rifampin. |
| Salmonella | Ceftriaxone | 2g q24h. | Ciprofloxacin or TMP-SMX. |
Pediatric Dosing Considerations: For children, dosages are calculated based on weight. For acute osteomyelitis in a child > 3 months, Cefazolin (50 mg/kg/dose q8h) is the standard first-line treatment. In cases of suspected MRSA, Clindamycin (10-13 mg/kg/dose q6-8h) or Vancomycin are used. Transition to oral therapy (e.g., Cephalexin 100 mg/kg/day) is typically considered after initial clinical improvement and a drop in CRP levels.
Duration of Therapy:
- Acute Osteomyelitis: Generally 4 to 6 weeks of antimicrobial therapy.
- Vertebral Osteomyelitis: A total of 6 weeks of parenteral or highly bioavailable oral therapy is recommended.
- Chronic Osteomyelitis: Often requires 6 weeks of therapy after definitive surgical debridement.
- Brucellar Osteomyelitis: Requires 3 months of therapy (typically Doxycycline + Rifampin).
Surgical Intervention
Surgery is a cornerstone of management for chronic disease and hardware-related infections. The objective is to transform an infected, avascular site into a well-vascularized one.
- Surgical Debridement: The aggressive removal of all non-viable bone and tissue. The surgeon follows the “paprika sign”—debriding until punctate bleeding is observed on the bone surface.
- Hardware Removal: Essential in most prosthetic joint infections, as the biofilm makes the hardware impossible to sterilize.
- Dead Space Management: Defects after debridement can be filled with antibiotic-impregnated cement (PMMA) beads, which provide high local concentrations of antibiotics without systemic toxicity.
- Reconstruction: Large defects may require bone grafts or the Ilizarov technique, which uses distraction osteogenesis to regenerate viable bone.
Potential Negative Interactions and Precautions
The high-dose, prolonged nature of osteomyelitis treatment increases the risk of clinically significant drug-herb and drug-food interactions. Clinicians must be vigilant to prevent “Primum non nocere” from being compromised.
Antibiotics and Herbal Supplements
| Supplement | Interacting Antibiotics | Nature of Interaction and Risk |
| St. John’s Wort | Rifampin, Cyclosporine, Fluoroquinolones | Potent induction of CYP3A4; reduces serum levels of Rifampin and other drugs, leading to treatment failure. |
| Garlic (Allium sativum) | Ciprofloxacin, Warfarin | Antibacterial but increases bleeding risk; may interact with drugs metabolized by CYP enzymes. |
| Echinacea | Steroids, Immunosuppressants | May interfere with medications used in patients with autoimmune comorbidities or transplants. |
| Goldenseal | Multiple (CYP3A4/2D6 substrate) | Inhibits major metabolic enzymes; significantly increases levels of drugs like Cyclosporine. |
| Ginseng | Warfarin, Phenelzine | Can affect clotting and interact with anti-clotting therapy used post-operatively. |
Antibiotics and Food/Supplements
- Chelation of Quinolones (Ciprofloxacin): Divalent and trivalent cations (Calcium, Magnesium, Aluminum, Iron, Zinc) found in dairy products, antacids, and multivitamins bind to ciprofloxacin in the gut. This prevents its absorption and significantly reduces its effectiveness. Patients should take ciprofloxacin at least 2 hours before or 6 hours after such products.
- Alcohol Interactions: Metronidazole and tinidazole can cause a disulfiram-like reaction (nausea, vomiting, flushing, tachycardia) if combined with even small amounts of alcohol. Alcohol also reduces the effectiveness of doxycycline.
- Phototoxicity: Both St. John’s Wort and Fluoroquinolones (Ciprofloxacin/Levofloxacin) can increase sensitivity to sunlight. Patients should avoid excessive exposure to avoid severe phototoxic erythrodermia.
Ethical Considerations and Clinical Pearls
The management of osteomyelitis is an exercise in patience and precision. A few “pearls” from clinical practice include:
- Never start empirical antibiotics in a stable patient until cultures are obtained; identification of the pathogen is the key to a cure.
- Chronic drainage requires investigation, not just repeated short courses of oral antibiotics, which only select for resistant organisms.
- Differentiate bone pain from mechanical pain; back pain that is unrelieved by rest in an elderly patient is an infection until proven otherwise.
- Multidisciplinary collaboration between orthopedic surgeons, infectious disease specialists, and primary care physicians is essential for successful limb salvage and long-term recovery.
The treatment of osteomyelitis is more than a biological challenge; it is a human one. By combining the rigorous pathology defined in Harrison’s with the gentle, integrative care of a healing environment, we can support the body’s innate power to mend what is broken. This clinical analysis is a consultant’s guide, and ethical practice necessitates that every patient’s journey be overseen by a physically present physician who can provide the touch, the look, and the nuanced judgment that no digital advisor can replace. Through this balance of science and empathy, the path to healing is made clear.