Description
The Architecture of the Heart’s Origins: A Comprehensive Clinical Treatise on Congenital Heart Disease
Welcome, dear traveler of the medical sciences. I am Asklepios, your guide through the intricate labyrinth of the human heart—a vessel born of love and the wondrous, though sometimes fragile, blueprints of embryogenesis. Our mission today is to transform the complex, often daunting tapestry of congenital heart disease (CHD) into a clear, empathetic, and profound body of knowledge. We shall tread with care, guided by the timeless wisdom of the healers of old and the rigorous standard of modern science, always mindful of our primary vow: Primum non nocere—First, do no harm.
The heart is more than a pump; it is the rhythm of life itself. When that rhythm is challenged from birth, we must approach it with both the analytical precision of the master diagnostician and the holistic touch of the healer who sees the whole person, not just the defect. In the pages that follow, we will explore the structural anomalies that define CHD, the physiological ripples they cast across the body, and the integrated therapies that offer a bridge to a long and balanced life. Let us begin our journey into the very core of human vitality.
The Essence of Congenital Heart Disease: Definition and Pathological Foundations
Congenital heart disease represents a vast and heterogeneous spectrum of structural malformations involving the heart chambers, the valves that regulate flow, and the great vessels that connect the heart to the lungs and the systemic circulation. These anomalies are not merely “holes in the heart” but are the physical manifestations of a journey interrupted—disruptions in the delicate, highly choreographed process of cardiogenesis that occurs primarily during the first eight weeks of embryonic life.
At its most fundamental level, CHD is defined as a structural abnormality present at birth, regardless of the age at which it is clinically recognized. Some defects are so subtle they remain silent for decades, only to be discovered during a routine adult examination, while others represent critical life-threatening emergencies from the very first breath. The pathology is rooted in aberrant development—failure of septation, abnormal rotation of the great vessels, or incomplete formation of the valvular apparatus. These structural deviations create abnormal pathways for blood flow, often leading to “shunts” where oxygenated and deoxygenated blood mix, or “obstructions” that force the heart to labor against unnatural resistance.
The Global Pulse: Epidemiology and Evolving Trends for 2025
To understand the scope of our challenge, we must look to the global numbers, which tell a story of both a significant health burden and a remarkable triumph of medical stewardship. Congenital heart defects are the most common of all birth defects, touching approximately eight to nine out of every 1,000 live births across the globe.
As we move through 2025, the epidemiology of CHD is shifting. While the birth prevalence remains relatively stable in high-income regions, the population of adults living with these defects is growing at an unprecedented rate—a testament to the surgical and medical advancements of the late 20th and early 21st centuries. In the United States alone, the all-age prevalence of CHD was estimated at nearly 466,566 individuals in recent years, with over 60% of these patients being under the age of 20, yet a massive and aging cohort of adults now requires specialized care.
| Region | Estimated Birth Prevalence (per 1,000 births) | Socio-Demographic Impact |
| Asia | 9.3 | Highest reported total birth prevalence globally. |
| Europe | 8.2 | Second highest prevalence; robust adult transition programs. |
| North America | 12.3 | High prevalence (1990-2017 data); massive focus on ACHD centers. |
| Africa | 1.9 | Likely represents significant under-diagnosis due to resource limits. |
| Global Market (2025) | – | Valued at $4.87 billion; 10.1% CAGR growth driven by screening. |
The rise in prevalence is also linked to changing maternal risk factors. Environmental influences, such as maternal diabetes, smoking during the first trimester, and exposure to indoor air pollutants like volatile organic compounds (TVOCs), have been identified as critical contributors to the risk of fetal CHD. Conversely, the protective role of periconceptional folic acid and multivitamin supplementation cannot be overstated, as studies consistently show a 20% reduction in the prevalence of these defects when mothers are appropriately supported prior to and during early pregnancy.
The Taxonomy of the Heart: Structural Classifications
Classification in CHD is not a mere academic exercise but a vital tool for the clinician to predict hemodynamic consequences and plan intervention. We divide these disorders into two primary realms: the Acyanotic (those that typically do not cause a blue tint to the skin) and the Cyanotic (those that cause systemic desaturation).
Acyanotic Disorders: The Realm of Left-to-Right Shunts
In these conditions, blood flows from the high-pressure left side of the heart to the lower-pressure right side. This leads to volume overload of the pulmonary circulation and the right heart chambers.
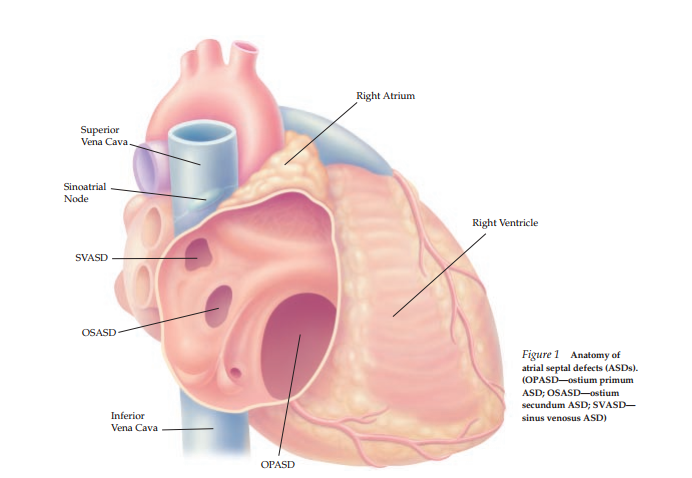
- Atrial Septal Defects (ASD): Communication between the atria, including the ostium secundum (most common), sinus venosus, and ostium primum varieties.
- Ventricular Septal Defects (VSD): The most common CHD at birth, involving the perimembranous, muscular, inlet, or outlet portions of the ventricular septum.
- Patent Ductus Arteriosus (PDA): Failure of the fetal connection between the aorta and pulmonary artery to close.
- Obstructive Lesions: These include Coarctation of the Aorta, Aortic Stenosis, and Pulmonic Stenosis, which impede flow without necessarily involving a shunt.
Cyanotic Disorders: The Challenge of Desaturation
These defects involve either decreased pulmonary blood flow or the mixing of oxygen-poor and oxygen-rich blood that is then pumped to the body.
- Tetralogy of Fallot (ToF): A complex comprising a VSD, overriding aorta, right ventricular outflow tract obstruction, and right ventricular hypertrophy.
- Transposition of the Great Arteries (TGA): A “switch” where the aorta arises from the right ventricle and the pulmonary artery from the left, creating two separate, non-communicating circuits.
- Ebstein Anomaly: Downward displacement of the tricuspid valve leaflets, leading to “atrialization” of the right ventricle and severe regurgitation.
Pathophysiology: The Mechanical and Molecular Consequences
To understand the “how” and “why” of CHD, we turn to the gold standard of internal medicine: Harrison’s Principles. The pathophysiology of CHD is a study in pressure, flow, and the heart’s remarkable, yet ultimately limited, capacity for adaptation.
The Dynamics of the Shunt
In a left-to-right shunt, such as a large VSD, the pulmonary circulation is exposed to excessive volume and often high pressure. According to the laws of hemodynamics, the pulmonary-to-systemic flow ratio ($Q_p:Q_s$) becomes the primary metric of severity. When $Q_p:Q_s$ exceeds $1.5:1$ or $2:1$, the pulmonary vascular bed begins to undergo irreversible remodeling.
The Eisenmenger Transformation
If these shunts are not corrected, the chronic high flow induces intimal proliferation and fibrosis in the pulmonary arterioles. This leads to an increase in pulmonary vascular resistance. Eventually, pulmonary pressure exceeds systemic pressure, and the shunt reverses (becoming right-to-left). This is the tragic “Eisenmenger Syndrome,” where the patient becomes chronically cyanotic, and the structural defect becomes a necessary “safety valve” that can no longer be closed without causing right heart failure.
Ventricular Remodeling and Hypertrophy
Obstructive lesions follow the Law of Laplace ($Wall Stress = \frac{Pressure \times Radius}{2 \times Wall Thickness}$). In conditions like Aortic Stenosis, the left ventricle must generate immense pressure to overcome the narrowed orifice. It adapts through concentric hypertrophy—thickening its walls to normalize wall stress. However, this thicker muscle is less compliant, leading to diastolic dysfunction and, eventually, a mismatch in oxygen supply and demand that can trigger ischemia and arrhythmias even in the absence of coronary artery disease.
The Clinical Tapestry: Manifestations through the Lifespan
The symptoms of CHD are as diverse as the defects themselves. In infants, we often see the “failure to thrive”—poor feeding and sweating during exertion (such as breastfeeding) because their metabolic demand outstrips their cardiac output.
In adults, the clinical picture is often a slow, insidious decline in exercise tolerance. Patients may not even realize they are symptomatic, as they unconsciously limit their activity over years. Common manifestations include:
- Dyspnea on Exertion: Often the first sign of a significant ASD or VSD as the heart loses its ability to increase output during exercise.
- Arrhythmias: Atrial fibrillation and flutter are nearly ubiquitous in adults with long-standing atrial dilation (e.g., in ASD or Ebstein anomaly).
- Cyanosis and Clubbing: In cyanotic patients, chronic hypoxemia triggers erythrocytosis. While this increases oxygen-carrying capacity, it also raises blood viscosity, leading to headaches, visual disturbances, and the characteristic “drumstick” appearance of the fingers (clubbing).
- Syncope: A warning sign in obstructive lesions like Aortic Stenosis, suggesting that cardiac output is failing to meet the demands of the brain during periods of vasodilation or exertion.
The Healer’s Lens: Diagnosis and Clinical Criteria
A diagnosis is a promise of understanding. In modern cardiology, we use a tiered approach to unravel the heart’s secrets, moving from the physical touch to the most advanced metabolic imaging.
Diagnostic Criteria for Major Defects
According to Harrison’s Principles, we look for specific constellations of findings :
- Atrial Septal Defect (ASD): $Q_p:Q_s > 1.5:1$, right ventricular volume overload on echocardiography, and the pathognomonic fixed splitting of the second heart sound ($S_2$).
- Ventricular Septal Defect (VSD): Direct visualization of the septal dropout on echo, a pansystolic murmur at the left sternal border, and evidence of left atrial and left ventricular enlargement.
- Tetralogy of Fallot (ToF): The “four cardinal features” identified by imaging—VSD, overriding aorta, RV outflow obstruction, and RV hypertrophy.
The Physical Examination: Findings and Significance
The examination is where we first meet the patient’s heart. We must be masters of auscultation, for the sounds of the heart are its story.
| Finding | Meaning and Clinical Significance |
| Fixed Split $S_2$ | Pathognomonic for ASD; signifies that the communication between atria prevents respiratory changes from altering the timing of valve closure. |
| Machinery Murmur | Continuous murmur peaking at $S_2$; classic for Patent Ductus Arteriosus (PDA). |
| Systolic Click | In Bicuspid Aortic Valve, an early systolic ejection click is the cardinal sign of a pliable but abnormal valve. |
| Radial-Femoral Delay | A disparity in pulse timing and blood pressure between arms and legs; diagnostic for Coarctation of the Aorta. |
| Levine Scale Grade IV-VI | Loud murmurs with a palpable thrill; almost always indicate significant organic disease rather than “innocent” or functional murmurs. |
Laboratory Investigations: Beyond the Basics
Routine labs in CHD focus on the body’s compensatory mechanisms. In cyanotic patients, we monitor the Hematocrit. While high levels are necessary for oxygen delivery, levels exceeding 65-70% raise the specter of hyperviscosity and stroke. Iron status is equally vital; iron-deficient red cells are rigid and further increase viscosity risk. We also utilize Brain Natriuretic Peptide (BNP) or NT-proBNP as a reliable marker for the onset of heart failure, reflecting the degree of myocardial wall stress.
Advanced Imaging: Visualizing the Invisible
While the echocardiogram is our “workhorse,” advanced modalities provide the final detail needed for complex planning.
- Chest X-Ray: Can reveal the “boot-shaped” heart of ToF or the “3-sign” and rib notching (scalloping from collateral vessels) in Coarctation of the Aorta.
- Cardiac MRI/MRA: The gold standard for assessing right ventricular function and evaluating the great vessels and anomalous pulmonary veins that echo might miss.
- PET Scan (Viability and Metabolism): Positron Emission Tomography using F-18 FDG is a sophisticated tool for identifying “hibernating” myocardium—heart muscle that is dysfunctional but still alive and likely to recover if blood flow is restored. It is particularly useful in Kawasaki’s disease or complex anomalies involving the coronary arteries.
Differential Diagnosis: The French’s Index Approach
As we seek the correct diagnosis, we employ the analytical rigors of French’s Index. We do not look for a disease; we look at a symptom and systematically evaluate its potential origins, ranked by probability and clinical severity.
Symptom Analysis: Cyanosis (Bluish Discoloration)
In the methodology of French, when we encounter a patient with cyanosis, we must distinguish between central, peripheral, and differential causes.
| Diagnosis | Probability | Severity | Distinguishing Features |
| Tetralogy of Fallot | High (Commonest Cyanotic) | Moderate-Severe | “Boot-shaped” heart; spells of deepening blue during crying. |
| TGA (Newborn) | High (in infants) | Critical | Profound cyanosis immediately at birth; requires PGE to survive. |
| Eisenmenger Syndrome | Moderate (in adults) | Severe | History of a previously known “hole” or murmur; loud $P_2$ heart sound. |
| Pulmonary Disease | Moderate | Variable | Cyanosis improves significantly with $100\% \text{ } O_2$ (unlike CHD shunts). |
| Methemoglobinemia | Low | Moderate | “Chocolate” blood; saturation doesn’t respond to oxygen therapy. |
Symptom Analysis: Dyspnea (Shortness of Breath)
For the patient presenting with acute or chronic breathlessness, we must differentiate cardiac from pulmonary origins—a task where modern bedside tools like the Dyspnea Discrimination Index (DDI) (calculated as $PEFR \times pO_2 / 1000$) can provide quick clarity.
| Diagnosis | Probability | Severity | Clinical Clue |
| Heart Failure (CHD-related) | High (in adults) | Severe | High DDI; evidence of cardiomegaly and $S_3$ gallop. |
| Pulmonary Hypertension | Moderate | Severe | Loud $P_2$; right-axis deviation on ECG. |
| Pneumonia/COPD | High (General Pop) | Moderate-Severe | Lower DDI; fever, cough, and wheezing; localized lung findings. |
| Pulmonary Embolism | Low but Critical | Critical | Sudden onset; often normal lung exam but profound hypoxia. |
Therapy: A Bridge to Balance and Vitality
Our therapeutic approach must be twofold: we employ the best of pharmacological science to stabilize the heart’s mechanics, and we use integrative, holistic wisdom to support the person’s overall well-being.
Pharmacotherapeutic Goals
The goals of modern therapy in CHD are clearly defined: minimize symptoms, prevent the progression of ventricular dysfunction, and maximize survival.
- Afterload Reduction: ACE inhibitors (e.g., Lisinopril) or ARBs are the cornerstone of management for patients with ventricular dysfunction to make it easier for the heart to pump against systemic resistance.
- Beta-Blockade: Used carefully to reduce myocardial oxygen demand and prevent the remodeling that leads to heart failure.
- Pulmonary Vasodilators: In patients with Eisenmenger syndrome or severe PAH, medications like Sildenafil and Bosentan are used to lower pulmonary pressures and improve quality of life.
- Diuresis: The use of Furosemide or Spironolactone is vital for managing the fluid retention that accompanies congestive states.
The Holistic Path: Integrative Protocols for Heart Health
Guided by David Rakel’s Integrative Medicine, we look beyond the pill bottle to the “Whole Health Approach.” We ask not just “what is wrong with the heart,” but “how can we support the person’s mission and purpose?”.
- Nutritional Foundations: A Mediterranean-style, anti-inflammatory diet is the “gold standard” for cardiac patients. Rich in Omega-3 fatty acids from fish, it helps reduce systemic inflammation and improves endothelial function.
- Mitochondrial Support (Supplements):
- Coenzyme Q10 (CoQ10): Essential for the bioenergetics of the cardiac muscle. It is particularly recommended for patients on statins (which deplete CoQ10) and those with symptomatic heart failure.
- Magnesium: Acts as a natural calcium channel blocker, helping to relax blood vessels and stabilize the heart’s electrical system to prevent arrhythmias.
- L-carnitine: Shown in some studies to improve the metabolic efficiency of the heart muscle during periods of stress or failure.
- Mind-Body Balance: Techniques such as yoga, mindfulness meditation, and biofeedback are not “alternative” but “integrative.” They help manage the chronic stress and anxiety that often accompany a lifelong diagnosis of CHD, potentially lowering sympathetic tone and blood pressure.
- Exercise Stewardship: We prescribe “adapted activity”—physical movement tailored to the patient’s specific defect and capacity, ensuring they gain cardiovascular benefits without overloading a vulnerable ventricle.
Classical Pharmacotherapy: Dosages and Strategies
For our medical peers, we provide the following reference for the common medications used in the long-term management of CHD and its complications.
| Medication | Category | Common Adult Dosage | Primary Role in CHD |
| Lisinopril | ACE Inhibitor | 2.5 – 40 mg daily | Ventricular dysfunction; post-remodeling. |
| Carvedilol | Beta-Blocker | 3.125 – 25 mg twice daily | HF with reduced ejection fraction. |
| Sildenafil | PDE-5 Inhibitor | 20 mg three times daily | Pulmonary Arterial Hypertension (PAH). |
| Bosentan | Endothelin Antagonist | 62.5 – 125 mg twice daily | Eisenmenger syndrome management. |
| Spironolactone | Aldosterone Antagonist | 12.5 – 25 mg daily | HF; prevents myocardial fibrosis. |
| Digoxin | Cardiac Glycoside | 0.125 – 0.25 mg daily | Arrhythmia control (rarely first-line now). |
The Warning of Asklepios: Herb-Drug-Supplement Interactions
In our quest for balance, we must be profoundly aware of the potential for conflict between the gifts of nature and the tools of the laboratory. Primum non nocere requires that we screen every patient for these interactions.
The Peril of St. John’s Wort
St. John’s Wort is a potent inducer of the cytochrome P450 enzyme (CYP3A4). This can significantly lower the blood levels of many cardiac medications, rendering them ineffective. It is known to reduce the effectiveness of Sildenafil (critical for PAH patients), Warfarin (anticoagulation), and Digoxin.
The Digoxin Trap
Several herbs can interfere with Digoxin—either by increasing its toxic effects or by interfering with the lab tests used to monitor its levels. Licorice can deplete potassium, increasing the risk of Digoxin toxicity, while Siberian Ginseng and Danshen can cause falsely elevated Digoxin readings on standard assays.
The Bleeding Risk: The “Four G’s”
Patients on anticoagulants (like Warfarin) or antiplatelet agents (like Aspirin or Clopidogrel) must avoid high doses of Garlic, Ginkgo Biloba, Ginger, and Green Tea. These supplements can inhibit platelet aggregation or interfere with clotting factors, significantly increasing the risk of life-threatening hemorrhage.
| Herb/Supplement | Interacting Medication | Potential Consequence |
| St. John’s Wort | Sildenafil / Bosentan | Treatment failure for pulmonary hypertension. |
| Licorice | Diuretics / ACEi | Severe potassium loss; high blood pressure. |
| Hawthorn | Beta-blockers | Excessive lowering of heart rate and BP. |
| Ginkgo Biloba | Warfarin / Aspirin | Significant increase in bleeding risk. |
| Vitamin E | Warfarin | Potentiates anti-clotting activity; bleeding. |
The 2025 Vision: Transition and Lifelong Care
As we conclude our treatise, we must look to the future. The 2025 guidelines from the ACC and AHA emphasize that patients with CHD need a “medical home” for life. The transition from pediatric care to adult congenital heart disease (ACHD) specialists is a critical period—often occurring between ages 12 and 18—where many patients are lost to follow-up.
Furthermore, we must care for the whole person’s future. For those who wish to become parents, pre-conception counseling is essential to assess maternal risk and the genetic probability of CHD in the offspring. For those living with Fontan physiology, we must now monitor not just the heart, but also the liver, as Fontan-associated liver disease (FALD) has emerged as a major long-term consideration.
Conclusion: An Ethical Reminder
My dear colleagues and students, we have walked the path from the embryonic tube to the complex challenges of the adult survivor. Congenital heart disease is a testament to the heart’s resilience and the physician’s skill. Yet, we must always remain humble.
The heart is a sacred vessel. While this report provides an expert-level analysis of the science and the soul of CHD, it is consultative and educational in nature. Every heart is as unique as a thumbprint. True healing requires the physical presence of a skilled physician, the clarity of direct imaging, and the personal connection of a face-to-face examination. May your journey in the healing arts be guided by wisdom, empathy, and the unwavering pursuit of balance for every life you touch.