Description
The Clinical Spectrum and Differential Diagnostic Paradigms of Loin Pain-Hematuria Syndrome
The loin pain-hematuria syndrome (LPHS) constitutes one of the most enigmatic and clinically challenging entities in the fields of nephrology and urology. Initially delineated in 1967 by Little and colleagues, who documented the cases of three young women suffering from recurrent, severe unilateral or bilateral flank pain accompanied by gross or microscopic hematuria, the syndrome has since evolved from a mere clinical curiosity into a recognized rare disease with profound implications for the quality of life of those affected. From a diagnostic perspective, LPHS is fundamentally a diagnosis of exclusion. The identification of this condition requires a meticulous and exhaustive clinical workup to eliminate a vast array of common and rare nephro-urological pathologies that present with similar symptomatology. Consequently, an expert understanding of its pathophysiology, clinical manifestations, and the nuances of its differential diagnostic markers is essential for any clinician managing chronic pelvic or flank pain.
Nosological Definition and Taxonomic Classification
Loin pain-hematuria syndrome is clinically defined by the chronic co-occurrence of severe, localized flank pain—which may be vacillating or persistent—and hematuria that originates from the glomerulus, rather than the lower urinary tract. The condition is recognized as a rare disease under the Rare Disease Act of 2002, with an estimated prevalence of approximately 0.012 percent in the general population.
In clinical practice, the syndrome is typically classified into two primary categories based on the etiology of the renal manifestations:
| Taxonomic Class | Clinical Definition and Pathological Context |
| Primary LPHS | This form occurs in the total absence of an identifiable, acquired underlying glomerular disease. It is often hypothesized to be linked to constitutive or genetic abnormalities of the glomerular basement membrane (GBM). |
| Secondary LPHS | This classification is applied when the clinical features of loin pain and glomerular bleeding occur concurrently with a documented acquired renal disease, such as Immunoglobulin A (IgA) nephropathy or various forms of vasculitis. |
Some academic circles further refine this classification based on the presence of anatomical or metabolic triggers. Type 1 LPHS has historically referred to cases where a potential “driver”—such as Nutcracker syndrome, non-obstructive nephrolithiasis, or polycystic kidney disease—is present. In contrast, Type 2 LPHS represents truly idiopathic cases where no morphological or metabolic abnormality can be uncovered despite exhaustive modern imaging and biochemical screening.
Epidemiological Profile and Patient Demographics
The epidemiology of LPHS is characterized by a high degree of specificity in patient demographics. The majority of affected individuals are young adults, with a mean age of onset of 31 years, although the clinical literature reports a range extending from the first to the sixth decades of life. There is a pronounced gender disparity; females represent between 70 and 80 percent of the patient population across most major clinical series. The factors driving this female predominance remain a subject of investigation, but researchers have pointed toward the fact that females statistically possess thinner glomerular basement membranes, which may increase the likelihood of glomerular capillary rupture under physiological stress.
Racial distribution data indicate that LPHS is overwhelmingly identified in White populations, accounting for approximately 94 percent of documented cases in major North American registries. Furthermore, a strong association exists between LPHS and a history of nephrolithiasis. Up to 50 percent of patients carry a history of passing kidney stones or exhibit renal calcifications (nephrocalcinosis) typical of stone disease, even if there is no active obstruction present during their characteristic pain crises.
The Pathophysiology of the Pain-Hematuria Cycle
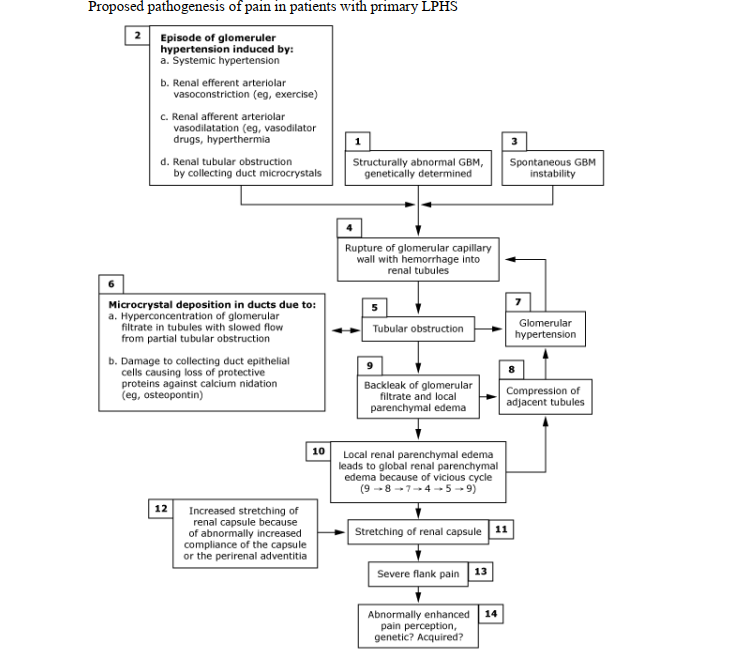
The transition from a microscopic glomerular event to a macroscopic clinical crisis in LPHS is explained through a multi-step pathological sequence. While various mechanisms such as renal vasospasm, renal artery disease, and coagulopathy were proposed in earlier decades, modern evidence favors a “vicious cycle” involving the glomerular basement membrane, tubular obstruction, and renal capsular tension.
Glomerular Basement Membrane Instability
The primary pathological insult in LPHS is hypothesized to be the structural instability of the glomerular basement membrane (GBM). Electron microscopy of renal biopsies from LPHS patients reveals that roughly 60 percent possess either abnormally thin or thick GBMs. These structural deviations predispose the capillary wall to rupture, particularly during episodes of glomerular hypertension. This hypertension may be systemic or localized, often induced by strenuous exercise, which causes renal efferent arteriolar vasoconstriction, or by vasodilator drugs and hyperthermia, which cause afferent arteriolar dilatation.
Hemorrhage and the Obstructive Sequence
When the glomerular capillary wall ruptures, red blood cells (RBCs) escape into the urinary space. This intra-nephron hemorrhage initiates a sequence of mechanical failures within the renal tubule:
| Sequence Step | Pathological Mechanism |
| Hemorrhage | Passage of RBCs through GBM gaps into the Bowman’s space and proximal tubule. |
| Cast Formation | RBCs aggregate within the tubular lumen, forming occlusive red cell casts. |
| Crystal Nidation | In patients with hypercalciuria or hyperuricosuria, microcrystals deposit in the collecting ducts, acting synergistically with RBC casts to enhance obstruction. |
| Loss of Protection | Damage to tubular epithelial cells leads to a loss of proteins like osteopontin, which normally inhibit crystal formation, further accelerating obstruction. |
Edema and Capsular Distension
The resulting tubular obstruction leads to an increase in intra-tubular pressure and the “backleak” of glomerular filtrate into the renal interstitium. This localized parenchymal edema compresses adjacent, previously patent tubules, creating a global edematous state within the renal parenchyma. Because the renal capsule is a relatively non-compliant fibrous structure, this parenchymal swelling results in significant stretching of the capsule. The capsule is rich in A-delta and C-fiber nociceptors, which transmit severe pain signals in response to mechanical distension. In some patients, there may also be a genetic or acquired hypersensitivity of these pain receptors, amplifying the perception of pain.
Clinical Manifestations and Patient Presentation
The clinical hallmark of LPHS is chronic, debilitating pain localized to the costovertebral angles. This pain is frequently described as throbbing or burning and is often of such intensity that it is compared to the most severe forms of renal colic.
Pain Characteristics and Triggers
Pain in LPHS is not a static symptom; it vacillates in intensity and is highly responsive to physical stimuli. The following clinical features are diagnostic clues:
- Localization: Typically unilateral at the onset, but longitudinal studies show that the majority of patients eventually develop bilateral symptoms.
- Exacerbation: Pain is significantly worsened by physical activity, vibrations (such as riding in a vehicle over rough roads), or even gentle percussion of the costovertebral angle.
- Positional Effects: Many patients report that lying in a supine position increases their flank pressure and pain intensity, often leading to significant sleep disturbances.
- Autonomic Involvement: Severe pain episodes are frequently associated with nausea and vomiting, which can complicates the management of oral medications.
Hematuria Patterns
The hematuria in LPHS is intrinsically linked to the pain cycle. While it may be microscopic between crises, it often transitions to gross hematuria during acute flares.
- Glomerular Morphology: Phase-contrast microscopy of the urine typically reveals dysmorphic red cells, specifically acanthocytes, and occasionally red cell casts.
- Clot Absence: Unlike hematuria from the bladder or ureter, LPHS-associated bleeding rarely results in the formation of blood clots, unless there is significant secondary tubular damage.
- Resolution Divergence: It is critical to note that the loin pain can persist for weeks or months even after the hematuria has macroscopically or microscopically cleared, likely due to residual parenchymal edema.
Typical Clinical Scenarios
Clinicians should be alert to three primary clinical presentations that are recurrent in the medical literature:
- The “Pyelonephritis” Mime: A young woman presents with recurring flank pain and microscopic hematuria. She is repeatedly treated with antibiotics for presumed pyelonephritis, despite persistently sterile urine cultures and the absence of pyuria.
- The Chronic Stone Patient: A patient with a documented history of urolithiasis continues to suffer from excruciating pain and hematuria long after the stone has been successfully passed or surgically removed and obstruction has been radiologically ruled out.
- The Athlete’s Hematuria: A high-performance athlete experiences gross hematuria and sudden, crippling flank pain during or immediately following intense physical exertion.
Diagnostic Evaluation: A Systematic Approach
The diagnosis of LPHS requires the fulfillment of specific criteria and the systematic exclusion of alternative causes of non-glomerular and glomerular bleeding.
Validated Diagnostic Criteria
The academic consensus suggests that a diagnosis of LPHS can be reached if the following parameters are satisfied :
- Symptom Duration: Pain and hematuria must be present for a period of at least six months.
- Pain Localization: Pain must be severe, localized at the costovertebral angles, and associated with costovertebral angle tenderness on examination.
- Hematuria Documentation: Hematuria ($> 5$ RBCs per high-power field) should be present in the vast majority of urinalyses.
- Negative Urologic Workup: Structural causes (stones, tumors, infections) must be definitively excluded.
- Absence of Obstruction: Lack of urinary tract obstruction must be confirmed by at least two separate imaging procedures during active pain episodes.
Laboratory Findings and Their Diagnostic Significance
Laboratory testing in LPHS focuses on demonstrating glomerular bleeding and metabolic predispositions while confirming the preservation of renal function.
| Parameter | Expected Finding | Clinical Significance |
| Urinalysis | Dysmorphic RBCs/Casts | Confirms glomerular origin of bleeding. |
| Serum Creatinine | Normal range ($0.7 – 1.3 \text{ mg/dL}$) | Differentiates LPHS from progressive nephropathies. |
| Proteinuria | $< 500 \text{ mg/day}$ | Higher levels suggest IgA or other primary glomerulopathies. |
| 24-hour Urine | Hypercalciuria/Hyperuricosuria | Identifies metabolic drivers of the pain-hematuria cycle. |
| Urine Culture | Persistently sterile | Essential to rule out chronic interstitial or recurrent infection. |
Advanced Imaging and Instrumental Studies
Radiological evaluation is the primary tool for the exclusion of competing diagnoses. In LPHS, these studies are typically unremarkable for gross pathology, which itself is a diagnostic clue.
- Renal Ultrasonography: Useful for initial screening to exclude hydronephrosis and large nephrolithiasis. Its lack of radiation makes it ideal for serial monitoring.
- Multidetector Computed Tomography (MDCT): CT urography is the gold standard for ruling out small, non-obstructing stones, renal masses, and morphological anomalies.
- Cystoscopy and Retrograde Pyelography: These are essential to exclude lower tract malignancy and ureteral strictures. In LPHS, the findings are classically negative.
- CT/MR Angiography: Necessary to evaluate for vascular anomalies such as Nutcracker syndrome or renal artery dissection.
The Role of Renal Biopsy and Histopathology
While not mandatory for all LPHS cases, renal biopsy becomes essential when there is clinical suspicion of an underlying acquired glomerular disease (secondary LPHS). Histological findings in primary LPHS are highly specific:
- Light Microscopy: Glomeruli typically appear normal. However, the presence of red cells or red cell casts in more than 3 percent of tubular cross-sections is a highly specific marker for glomerular hematuria in these patients.
- Immunofluorescence: Classically negative for immune deposits, helping to distinguish LPHS from IgA nephropathy.
- Electron Microscopy: The definitive tool for identifying structural GBM abnormalities. Approximately 60 percent of specimens will show either significant thinning ($< 250 \text{ nm}$) or excessive thickening of the basement membrane.
Differential Diagnosis: Navigating the Complex Clinical Landscape
The differential diagnosis of loin pain and hematuria is arguably the most critical section for the clinician. Given that LPHS is an exclusion diagnosis, a systematic review of its “mimics” is paramount.
Obstructing and Non-Obstructing Urolithiasis
The most common cause of flank pain and hematuria is nephrolithiasis. While LPHS patients often have a history of stones, the primary distinction is the absence of obstruction during the pain episode.
- Diagnostic Clue: In LPHS, CT imaging during a pain crisis will show no evidence of a stone in the ureter or hydroureteronephrosis.
- Metabolic Overlap: There is a significant overlap in risk factors; hypercalciuria and hyperuricosuria contribute to both conditions, but in LPHS, the pain is likely driven by microcrystal-induced tubular edema rather than macroscopic mechanical obstruction.
IgA Nephropathy (Berger’s Disease)
IgA nephropathy is the most common primary glomerulonephritis worldwide and a frequent cause of secondary LPHS.
- Similarities: Both can present with gross hematuria following upper respiratory infections or exercise and associated flank pain.
- Differences: IgA nephropathy is more likely to be associated with significant proteinuria ($> 1 \text{ g/day}$), hypertension, and a gradual decline in the glomerular filtration rate (GFR). Renal biopsy with positive immunofluorescence for IgA is definitive.
Nutcracker Syndrome (Left Renal Vein Entrapment)
Nutcracker syndrome (NCS) is perhaps the most significant “vascular mimic” of LPHS, particularly because it predominantly affects young, thin females.
- Anatomical Mechanism: The left renal vein (LRV) is compressed between the aorta and the superior mesenteric artery (anterior NCS) or between the aorta and the vertebral column (posterior NCS).
- Clinical Differentiation: NCS pain is strictly unilateral (left-sided) and may be associated with symptoms of pelvic congestion (dyspareunia, dysmenorrhea) or orthostatic proteinuria.
- Imaging Markers: CT showing a “beak sign” at the LRV and a pressure gradient of $\ge 3 \text{ mm Hg}$ between the LRV and the IVC are diagnostic of NCS, whereas LPHS will show a patent LRV without a significant gradient.
Thin Basement Membrane Nephropathy (TBMN)
TBMN, or benign familial hematuria, is a common genetic condition often confused with LPHS.
- Diagnostic Distinction: TBMN is usually a lifelong, asymptomatic condition characterized by persistent microscopic hematuria. It rarely causes loin pain. When a patient with biopsy-proven TBMN develops severe loin pain, they are categorized as having LPHS secondary to TBMN.
Malignancy and Structural Anomalies
Renal cell carcinoma (RCC) and urothelial carcinoma must always be considered in the differential of any patient with hematuria, regardless of age.
- RCC: Usually presents as a solid, enhancing mass on CT.
- Urothelial Cancer: May present as a filling defect in the renal pelvis or ureter.
- ADPKD: Pain is typically caused by cyst hemorrhage, rupture, or infection, and the diagnosis is confirmed by the presence of multiple bilateral cysts on imaging.
Rare Vascular and Embolic Events
- Renal Infarction: Typically sudden onset in patients with risk factors like atrial fibrillation. CT with IV contrast shows a “wedge-shaped” non-enhancing defect.
- Renal Artery Dissection: Associated with fibromuscular dysplasia (FMD) or trauma; requires CT or MR angiography for visualization.
- Renal Vein Thrombosis: More common in patients with nephrotic-range proteinuria; presents with acute flank pain and an enlarged, edematous kidney on ultrasound.
Differential Diagnostic Comparison Table
| Condition | Primary Side | Hematuria Type | Imaging Key | Proteinuria |
| LPHS | Uni/Bilateral | Glomerular | Normal/GBM anomaly | Minimal |
| Nutcracker | Left only | Non-Glom/Glom | LRV compression | Orthostatic |
| IgA Nephropathy | Bilateral | Glomerular | Normal | Significant |
| Urolithiasis | Usually Uni | Non-Glomerular | Stone/Hydronephrosis | None |
| Renal Infarction | Usually Uni | Non-Glomerular | Wedge defect on CT | None |
Psychological and Somatoform Considerations
A significant debate exists in the literature regarding the psychological profile of LPHS patients. Because the pain is severe and standard imaging is often negative, a psychiatric component has been frequently proposed.
- Somatoform Pain Disorder: Some studies have noted that LPHS patients are more likely to have medically unexplained somatic symptoms or a history of adverse psychological events preceding the onset of pain.
- Chronic Pain Syndrome: On the contrary, many experts believe the psychiatric disturbances (depression, anxiety) are secondary to the burden of living with a chronic, misunderstood, and incapacitating pain syndrome. This is supported by the fact that patients often experience a reversal of psychiatric symptoms once effective pain control is achieved through medical or surgical means.
Principles of Management and Therapeutic Goals
As per the differential-diagnostic focus of this article, therapeutic interventions are summarized here primarily to contextualize the clinical management strategies and their role in the diagnostic trial.
Pharmacotherapeutic Goals
Management aims to minimize glomerular bleeding and stabilize chronic pain.
- ACE Inhibition: The use of medications like lisinopril or valsartan is based on the rationale that lowering intraglomerular pressure reduces the frequency of GBM rupture and hematuria.
- Analgesia: A careful, stepwise approach using non-opioids followed by opioids for exacerbations is standard. The involvement of a multidisciplinary Pain Service is essential to prevent opioid-induced hyperalgesia and addiction.
- Metabolic Management: For patients with hypercalciuria, thiazide diuretics may reduce microcrystal-induced tubular damage.
Invasive and Experimental Procedures
For refractory cases, various invasive options exist, although their success is variable:
- Renal Denervation: Surgical or endovascular ablation of the renal nerves can provide temporary relief, but pain often recurs due to re-innervation.
- Intrathecal Opioid Pumps: In some centers, implantable pumps for the delivery of morphine or hydromorphone into the spinal space have been effective in reducing hospitalizations.
- Renal Autotransplantation: This major surgical procedure involves harvesting the kidney and re-implanting it in the iliac fossa to achieve complete denervation. While reported as a “last resort” with a 70-76% success rate in some series, it carries significant surgical risks and a notable rate of pain recurrence.
Potential Adverse Interactions
Clinicians must be vigilant regarding herb-drug interactions, particularly in patients on polypharmacy for pain:
- NSAID Risk: NSAIDs like ibuprofen can reduce renal perfusion and should be used with extreme caution or avoided in LPHS, especially in patients already on ACE inhibitors.
- St. John’s Wort: This herbal supplement significantly induces CYP3A4, which can accelerate the metabolism of opioids such as oxycodone and fentanyl, leading to inadequate analgesia.
- High-Potassium Herbs: Supplements like Alfalfa or Dandelion should be avoided as they can interfere with electrolyte balance in patients on blood pressure medications.
Natural History and Prognosis
The natural history of LPHS is characterized by high morbidity but excellent long-term renal survival. There is no evidence that LPHS leads to end-stage renal disease (ESRD) or increases mortality. In many patients (up to 50 percent), the syndrome resolves spontaneously over several years, particularly as they approach the sixth decade of life. A potential mechanism for this resolution is that the glomeruli most prone to bleeding eventually become non-functional through localized tubular damage; if this process involves only a small percentage of total nephrons, the pain resolves while the overall kidney function remains stable.
In conclusion, Loin pain-hematuria syndrome remains a diagnosis of profound exclusion that requires the clinician to be both a meticulous investigator and a supportive advocate for the patient. By systematically ruling out structural, vascular, and acquired glomerular diseases, and by understanding the microscopic clues provided by renal biopsy and 24-hour metabolic screening, medical professionals can accurately identify this challenging condition and provide the multidisciplinary care required to manage its complex symptom profile.