Description
Clinical Electrophysiology, Differential Diagnosis, and Therapeutic Stratification of Wolff-Parkinson-White Syndrome
Executive Summary of Pre-excitation Phenotypes
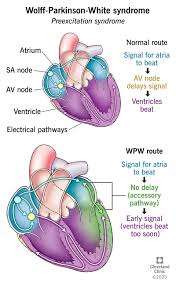
The Wolff-Parkinson-White (WPW) syndrome represents the most clinically significant form of ventricular pre-excitation, a condition where the cardiac ventricles are activated by an impulse originating in the atria earlier than would be expected if the impulse traveled solely through the physiological atrioventricular (AV) conduction system. This early activation is mediated by an anomalous anatomical bridge known as an accessory pathway (AP) or the “bundle of Kent”. Since its initial description in 1930, our understanding of this syndrome has evolved from a benign electrocardiographic curiosity to a potentially life-threatening condition characterized by paroxysmal supraventricular tachycardias and a risk of sudden cardiac death (SCD).
The prevalence of the WPW pattern—the characteristic ECG findings in an asymptomatic individual—is estimated at 0.1% to 0.3% of the general population. However, the progression from the “pattern” to the “syndrome” occurs when these individuals develop symptoms related to tachyarrhythmias. The clinical spectrum is broad, necessitating a sophisticated diagnostic approach that combines meticulous surface electrocardiography, clinical risk stratification, and, in many cases, invasive electrophysiological evaluation.
Pathophysiology and Structural Basis of Pre-excitation
In the electrophysiologically normal heart, the fibrous skeleton provides electrical insulation between the atria and the ventricles, with the AV node serving as the solitary conduit for impulse transmission. The AV node possesses the unique property of decremental conduction, introducing a physiological delay ($PR$ interval) that allows for optimal ventricular filling following atrial systole. In patients with WPW syndrome, one or more accessory pathways—remnants of incomplete embryological separation of the atrioventricular myocardium—cross this insulating barrier.
Most accessory pathways are located along the mitral or tricuspid annulus, though they can also be found in the septal regions. These pathways typically lack the decremental conduction properties of the AV node, allowing for rapid, non-delayed impulse transmission from the atria to the ventricles. Consequently, the ventricular myocardium is activated by two simultaneous wavefronts: one traveling down the accessory pathway (causing pre-excitation) and another following the normal AV nodal-His-Purkinje system.
Classification of Accessory Pathways and Vectors
Accessory pathways are classified by their location and their ability to conduct impulses in an antegrade or retrograde direction. While most conduct bidirectionally, “concealed” pathways conduct only in a retrograde fashion (ventricle to atrium). These pathways do not produce pre-excitation during sinus rhythm but can serve as a limb for re-entrant tachycardias.
| Pathway Type | Conduction Direction | Sinus ECG Findings | Clinical Implications |
| Overt Pathway | Bidirectional | Short $PR$, Delta wave, Wide $QRS$ | Risk of pre-excited AF and AVRT |
| Concealed Pathway | Retrograde Only | Normal $PR$, No Delta wave | Risk of orthodromic AVRT only |
| Antegrade Only | Antegrade Only | Short $PR$, Delta wave, Wide $QRS$ | Rare; risk of pre-excited AF |
The anatomical localization of the pathway determines the initial vector of ventricular depolarization, which is reflected in the polarity of the delta wave across the 12-lead ECG. This is traditionally categorized into two main types, though more precise localization algorithms exist:
- Type A (Left-sided pathway): The delta wave is positive in all precordial leads ($V_1$–$V_6$), and the $R/S$ ratio in lead $V_1$ is greater than 1.
- Type B (Right-sided pathway): The delta wave is negative or isoelectric in leads $V_1$ and $V_2$, with a precordial transition occurring later (typically after $V_2$).
Genetic Factors and Associated Structural Anomalies
While most cases are sporadic, approximately 3.4% of first-degree relatives of affected individuals may also exhibit the syndrome. A familial form of WPW syndrome has been associated with mutations in the $PRKAG2$ gene, which regulates adenosine monophosphate-activated protein kinase. This mutation often leads to a combination of ventricular pre-excitation, conduction system disease, and hypertrophic cardiomyopathy.
Furthermore, WPW syndrome is frequently associated with Ebstein’s anomaly, a congenital malformation of the tricuspid valve. In these patients, multiple accessory pathways are more common, and the pathways are typically located in the right-sided or posterior-septal regions.
Clinical Manifestations and Symptomatology
The clinical presentation of WPW syndrome is diverse, spanning from the asymptomatic “WPW pattern” found incidentally on a routine ECG to catastrophic sudden cardiac arrest.
Adult and Adolescent Presentation
Palpitations are the most common symptom, typically characterized by a sudden onset and offset of a rapid, regular or irregular heartbeat. These episodes can be debilitating and are often accompanied by:
- Dizziness and Lightheadedness: Resulting from transient hypotension during rapid heart rates.
- Dyspnea and Anxiety: Often triggered by the physical sensation of the racing heart.
- Chest Pain or Tightness: Which may mimic anginal pain, especially if the heart rate is high enough to compromise myocardial perfusion.
- Syncope or Near-syncope: A critical finding that often suggests rapid conduction over the accessory pathway (SPERRI $< 250 \text{ ms}$) or the development of atrial fibrillation with a fast ventricular response.
Pediatric and Infant Considerations
In infants, the clinical signs are subtle and require high clinical suspicion. Presentation may include:
- Irritability and Restlessness: The infant may be inconsolable during tachycardic episodes.
- Poor Feeding and Diaphoresis: Often occurring during feedings, which serve as a form of physical stress.
- Cyanosis or Pallor: Specifically, a blue or grayish hue to the skin, lips, and nails, indicating congestive heart failure or poor perfusion.
- Rapid Breathing (Tachypnea): A compensatory mechanism for reduced cardiac output.
For children aged 5 years or older, catheter ablation is increasingly recognized as the treatment of choice due to its high efficacy and favorable cost-effectiveness compared to long-term pharmacological management or surgery.
Diagnostic Electrocardiography
The diagnosis of WPW pattern is essentially an electrocardiographic one, characterized by the hallmark “fusion” complex during sinus rhythm.
Standard ECG Criteria
The following three criteria are mandatory for the diagnosis of overt ventricular pre-excitation:
- Shortened $PR$ Interval: A duration of $< 120 \text{ ms}$ (and frequently $< 100 \text{ ms}$).
- Delta Wave: A slurred, slow upstroke of the initial portion of the $QRS$ complex, representing early depolarization of the ventricular muscle at the site of the accessory pathway insertion.
- Broad $QRS$ Complex: A duration typically $> 110\text{–}120 \text{ ms}$, resulting from the combination of early pre-excitation and the subsequent normal activation wavefront.
Secondary $ST$-segment and $T$-wave changes are frequently observed. These repolarization abnormalities are usually “discordant,” meaning they are directed opposite to the major deflection of the $QRS$ complex and the delta wave.
Minimal Pre-excitation and Subtle Findings
In cases where the accessory pathway is located far from the sinus node (e.g., a left lateral pathway) or when the AV nodal conduction is very rapid, pre-excitation may be “minimal”. In such instances, the $PR$ interval may be at the lower limit of normal ($120 \text{ ms}$), and the delta wave may be barely visible.
To identify minimal pre-excitation, clinicians should look for:
- Absence of a septal $Q$ wave in lead $V_6$: This is a sensitive indirect sign of pre-excitation, as early activation of the lateral wall by an accessory pathway obliterates the normal left-to-right septal depolarization.
- Variation of the $PR$ interval: A difference of $> 30 \text{ ms}$ between the longest and shortest $PR$ intervals on a single 12-lead ECG may suggest an accessory pathway.
Localization by Delta Wave Polarity
The 12-lead ECG can be used to approximate the location of the accessory pathway, which is essential for planning catheter ablation.
| Lead | Left Lateral Pathway | Right Free-Wall Pathway | Septal Pathway |
| $V_1$ | Positive Delta ($R/S > 1$) | Negative/Isoelectric Delta | Positive Delta |
| II, III, aVF | Positive Delta | Negative Delta (if posterior) | Positive or Negative |
| I, aVL | Negative Delta | Positive Delta | Positive Delta |
Differential Diagnosis and Electrophysiological Mimicry
The differential diagnosis of WPW syndrome involves two main challenges: distinguishing it from other pre-excitation syndromes and differentiating WPW-related tachycardias from other causes of wide-complex tachycardia.
Comparison with Other Pre-excitation Syndromes
Several variants of accessory tracts exist, each with unique electrocardiographic profiles.
Lown-Ganong-Levine (LGL) Syndrome
Historically linked to “James fibers” that bypass the AV node by connecting the atria directly to the bundle of His.
- ECG Criteria: Short $PR$ interval ($< 120 \text{ ms}$) but a normal $QRS$ duration and no delta wave.
- Clinical Significance: The existence of LGL as a distinct structural entity is controversial; many cases are now thought to represent enhanced AV nodal conduction.
Mahaim-Type Pre-excitation
Mahaim fibers typically represent right-sided atriofascicular or nodoventricular tracts that conduct only in an antegrade direction and exhibit decremental properties.
- ECG Criteria: Normal or near-normal $PR$ interval ($0.12$–$0.20 \text{ s}$) with a subtle delta wave during sinus rhythm.
- Tachycardia Morphology: Typically produces an antidromic AVRT with a Left Bundle Branch Block (LBBB) pattern and a superior axis.
Fasciculoventricular Pathways
These are rare pathways connecting the His bundle or the fascicles directly to the ventricular myocardium.
- ECG Criteria: Fixed pre-excitation that does not change with exercise or vagal maneuvers, often with a normal $PR$ interval.
- Clinical Significance: Generally considered benign as they do not typically participate in re-entrant tachycardias.
Pseudo-Infarction Patterns
Ventricular pre-excitation can mimic myocardial infarction, leading to unnecessary diagnostic evaluations or treatments.
- Inferior Infarction: Negative delta waves in leads II, III, and aVF can resemble pathological $Q$ waves.
- Lateral Infarction: Negative delta waves in leads I and aVL.
- Posterior Infarction: A dominant $R$ wave in lead $V_1$, common in left-sided pathways, can be mistaken for posterior myocardial infarction.
Wide Complex Tachycardia (WCT) Differentiation
Distinguishing antidromic AVRT or pre-excited AF from Ventricular Tachycardia (VT) is a paramount diagnostic necessity.
The Brugada Algorithm for VT vs. SVT with Aberrancy
- Absence of an RS complex in all precordial leads? If yes, diagnosis is VT.
- RS interval > 100 ms in any precordial lead? (Measured from the onset of the $R$ wave to the nadir of the $S$ wave). If yes, diagnosis is VT.
- Atrioventricular (AV) dissociation? If yes, diagnosis is VT.
- Morphological criteria for VT in $V_1$–$V_2$ and $V_6$? (e.g., taller left “rabbit ear” in $V_1$, $QS$ in $V_6$). If yes, diagnosis is VT.
Pre-excited AF vs. Polymorphic VT
Pre-excited AF (AF in the setting of WPW) presents as a “fast, broad, and irregular” tachycardia.
- Clues for Pre-excited AF: Irregularly irregular rhythm, very high rates (sometimes $> 250 \text{ bpm}$), and varying $QRS$ morphologies as the degree of fusion changes.
- Treatment: Immediate cardioversion is mandatory if unstable. Avoid all AV nodal blockers.
Therapeutic Management and Risk Stratification
The management of WPW syndrome is stratified based on symptoms, the nature of the arrhythmia, and the assessed risk of sudden cardiac death.
Acute Emergency Management
Hemodynamically Unstable Patients
Immediate synchronized electrical cardioversion is indicated for any patient with WPW who is unstable (hypotension, shock, pulmonary edema, or altered mental status).
Hemodynamically Stable Patients
- Orthodromic AVRT (Narrow QRS): Initial management includes vagal maneuvers (Valsalva or carotid sinus massage). If unsuccessful, intravenous adenosine is the drug of choice.
- Pre-excited AF or Antidromic AVRT (Wide QRS): Intravenous Procainamide or Ibutilide are the agents of choice as they primarily target the accessory pathway.
Absolute Contraindications (The “ABCD” of WPW)
In the presence of an overt accessory pathway and an atrial arrhythmia (AF or Flutter), the following agents must be avoided:
- Adenosine
- Beta-blockers
- Calcium channel blockers (Verapamil, Diltiazem)
- Digoxin
These drugs block the AV node, which can facilitate conduction exclusively through the accessory pathway, leading to ventricular rates so rapid that they degenerate into ventricular fibrillation.
Chronic Pharmacological Management
Pharmacotherapy is often utilized as a bridge to ablation or in cases where ablation is refused or contraindicated.
| Drug | Class | Mechanism | Adult Dose | Key Side Effects |
| Flecainide | IC | Sodium channel blockade | $50\text{–}150 \text{ mg}$ BID | Dizziness, Blurred vision |
| Sotalol | II/III | Beta-block + $K^+$ channel block | $80\text{–}160 \text{ mg}$ BID | Bradycardia, Long $QT$ |
| Amiodarone | III | Multiple ion channel block | $200\text{–}400 \text{ mg}$ daily | Thyroid/Lung toxicity |
Definitive Therapy: Catheter Ablation
Radiofrequency (RF) catheter ablation has a success rate of over 90–95% and is considered the definitive cure for WPW syndrome.
- Procedure: Catheters are guided via the venous system to map the accessory pathway. RF energy or cryoablation is then used to permanently disrupt the pathway.
- Indications: Symptomatic tachyarrhythmias, high-risk asymptomatic individuals (high-risk professions), or patients with Ebstein’s anomaly.
- Complications: Although rare ($< 1\%$), complications can include heart block, cardiac tamponade, or vascular injury.
Integrative and Holistic Approaches in the Therapeutic Plan
In the modern management of cardiac rhythm disorders, integrative medicine serves as a complementary component within a comprehensive treatment plan. It is not intended to replace standard medical or interventional therapies but rather to optimize the patient’s overall physiological and psychological state, potentially reducing the frequency of symptomatic episodes.
Nutritional and Metabolic Stabilization
Optimal electrolyte balance is critical for cardiac electrical stability.
- Magnesium: Often referred to as “nature’s calcium channel blocker,” magnesium is essential for maintaining the resting membrane potential of cardiac myocytes. Deficiency can lower the threshold for triggered activity and re-entry.
- Omega-3 Fatty Acids: High-quality fish oils have been shown to stabilize cardiac cell membranes and exert a mild anti-inflammatory effect, which may be beneficial in reducing the trigger events for AF.
- Coenzyme Q10 (CoQ10): Particularly relevant if there is concomitant structural heart disease or cardiomyopathy, as it supports mitochondrial function and ATP production in the myocardium.
Botanical Considerations and Safety
Some herbal remedies can support cardiac health but must be monitored for significant interactions with conventional antiarrhythmics.
- Hawthorn (Crataegus): Known for its ability to improve coronary circulation and provide mild support for cardiac output. In the context of WPW, it should be viewed as a general cardiac tonic rather than a rhythm-specific treatment.
- Interactions with Antiarrhythmics: St. John’s Wort should be avoided by patients on amiodarone as it may induce hepatic enzymes and reduce the drug’s effectiveness. Similarly, grapefruit juice inhibits the metabolism of amiodarone, potentially leading to toxic levels and proarrhythmic risk.
Lifestyle and Autonomic Balance
Chronic stress and excessive sympathetic activation can lower the refractory period of both the AV node and the accessory pathway, making re-entrant tachycardias more likely.
- Stress Reduction: Practices such as meditation, yoga, and heart-rate variability (HRV) biofeedback can help balance the autonomic nervous system.
- Trigger Avoidance: Patients should strictly limit the use of stimulants, including caffeine, nicotine, and alcohol, as these are well-documented triggers for paroxysmal SVT in WPW.
Risk Stratification and Long-Term Outlook
The risk of sudden cardiac death in WPW syndrome is low ($0.1\%$ to $0.4\%$ annually) but significant, as cardiac arrest is the first manifestation of the disease in nearly half of those who suffer from it.
High-Risk Features for Sudden Death
Identifying the “needle in the haystack” remains a challenge. High-risk features include:
- SPERRI < 250 ms: A shortest pre-excited RR interval during AF of less than $250 \text{ ms}$ is the most predictive marker for risk of VF.
- Multiple Accessory Pathways: Common in Ebstein’s anomaly.
- Male Gender and Age < 30 years: Statistical risk is higher in this demographic.
- History of AF or Syncope: Symptomatic patients are at higher risk than those with a simple WPW pattern.
Prognosis and Post-Ablation Care
Most patients with WPW syndrome lead normal, active lives after successful ablation. The recurrence rate after a successful first procedure is low, and second procedures are usually definitive. For asymptomatic patients who choose conservative management, regular follow-up with a 12-lead ECG every 6 months to monitor $QRS$ width and corrected $QT$ interval is prudent.
Conclusion
The Wolff-Parkinson-White syndrome is a cornerstone of clinical electrophysiology, demonstrating the profound impact that a tiny anatomical bridge can have on cardiac rhythm and patient survival. Diagnosis rests on the triad of short $PR$, delta wave, and wide $QRS$, but the clinician must remain vigilant in differentiating it from LGL, Mahaim fibers, and more sinister wide-complex tachycardias like VT.
Management has been revolutionized by catheter ablation, offering a definitive cure for over $90\%$ of patients. In the acute setting, the avoidance of AV nodal blockers during pre-excited AF remains a life-saving principle. While integrative therapies like magnesium and stress management provide valuable metabolic and autonomic support, they remain an adjunctive part of a comprehensive plan led by interventional electrophysiology. Through meticulous risk stratification and a multidisciplinary approach, the threat of malignant arrhythmias in this population can be almost entirely neutralized.