Description
Comprehensive Analysis of Hemostatic Abnormalities in Preeclampsia: Pathophysiological Mechanisms, Clinical Diagnostics, and Therapeutic Implications
The Hemostatic Paradigm in Gestational Physiology
The progression of a normal pregnancy necessitates a profound systemic recalibration of the hemostatic system, evolving into a physiological state of hypercoagulability. This transition is primarily characterized by a significant increase in the concentration of most procoagulant factors, a concomitant reduction in the activity of certain natural anticoagulants, and a relative suppression of the fibrinolytic system. Such adaptations represent an evolutionary mechanism designed to minimize the catastrophic risk of postpartum hemorrhage during the third stage of labor and placental separation. However, this hypercoagulable milieu simultaneously increases the risk of thromboembolic events, which are further exacerbated when pathological conditions such as preeclampsia manifest.
Preeclampsia is a complex multisystemic disorder, occurring in 5% to 10% of hypertensive pregnancies, and remains a leading cause of maternal and neonatal morbidity and mortality worldwide. It is clinically defined by the new onset of hypertension (systolic $\geq 140$ mmHg or diastolic $\geq 90$ mmHg) and typically proteinuria ($\geq 300$ mg in a 24-hour collection) after the 20th week of gestation. In more severe presentations, the diagnosis can be established in the absence of proteinuria if there is evidence of end-organ dysfunction, such as thrombocytopenia, impaired liver function, or renal insufficiency. The hemostatic abnormalities reported in preeclampsia are not merely secondary complications but are intrinsic to the disease’s core pathophysiology, involving a self-perpetuating cycle of endothelial activation, platelet consumption, and localized or systemic intravascular coagulation.
| Hemostatic Variable | Normal Pregnancy | Preeclampsia |
| Fibrinogen | Increased (up to 100%) | Normal to Decreased (Consumption) |
| Von Willebrand Factor (VWF) | Increased | Significantly Increased |
| Antithrombin III | Stable | Decreased |
| Protein C | Stable | Decreased |
| Protein S (Free) | Decreased | Further Decreased |
| Plasminogen Activator Inhibitor-1 (PAI-1) | Increased | Marked Increase |
| Plasminogen Activator Inhibitor-2 (PAI-2) | Increased | Decreased |
| Thrombin-Antithrombin (TAT) Complexes | Increased | Significantly Increased |
| Platelet Count | Stable to Slightly Decreased | Often Significantly Reduced |
| Fibrin D-dimer | Increased | Significantly Increased |
Pathophysiological Foundations: The Two-Stage Model and Angiogenic Imbalance
The prevailing understanding of preeclampsia pathophysiology centers on a “two-stage model”. Stage I is characterized by defective trophoblast invasion of the uterine spiral arteries early in the first trimester. In a healthy pregnancy, trophoblasts remodel these narrow, high-resistance vessels into wide, low-resistance channels to ensure adequate placental perfusion. In preeclampsia, this remodeling is incomplete, leaving the arteries muscular and responsive to vasomotor influences, which leads to chronic placental ischemia and hypoxia.
Stage II involves the maternal systemic response to placental distress. The ischemic placenta releases a variety of soluble factors into the maternal circulation, including antiangiogenic proteins, pro-inflammatory cytokines, and syncytiotrophoblast microparticles. These factors trigger widespread maternal endothelial dysfunction, which is the direct cause of the hypertension and the multisystemic hemostatic failures observed.
The Role of sFlt-1 and sEng in Endothelial Activation
A pivotal mechanism in Stage II is the imbalance of angiogenic factors. The preeclamptic placenta overproduces soluble fms-like tyrosine kinase-1 (sFlt-1) and soluble endoglin (sEng). sFlt-1 acts as a decoy receptor, binding to and neutralizing vascular endothelial growth factor (VEGF) and placental growth factor (PlGF) in the maternal circulation. Because VEGF and PlGF are critical for maintaining the health and normal anticoagulant properties of the endothelium, their neutralization leads to a loss of endothelial integrity.
Healthy endothelial cells produce nitric oxide (NO) and prostacyclin ($PGI_2$), which are potent vasodilators and inhibitors of platelet aggregation. In preeclampsia, the antiangiogenic state suppresses the production of these protective molecules. Concurrently, there is an increased production of thromboxane $A_2$ ($TXA_2$), a potent vasoconstrictor and platelet aggregator. The resulting imbalance in the $PGI_2:TXA_2$ ratio promotes a pro-aggregatory and pro-thrombotic environment.
Oxidative Stress and Lipid Peroxidation
Placental ischemia also induces significant oxidative stress. The generation of reactive oxygen species (ROS) and lipid peroxidation products directly damages the maternal vascular endothelium. Research has indicated that lipid peroxidation degradation products are significantly increased in the placentas of preeclamptic women, contributing to a state of systemic inflammation. Furthermore, abnormal lipid profiles—characterized by elevated total cholesterol, triglycerides, and low-density lipoprotein (LDL) along with decreased high-density lipoprotein (HDL)—are strongly associated with endothelial dysfunction and atherosclerotic-like changes in the placental vasculature.
| Lipid Parameter | Mean ± SD (Control) | Mean ± SD (Preeclampsia) | Diagnostic Cut-off (mg/dL) |
| Total Cholesterol | $187.20 \pm 27.30$ | $238.46 \pm 38.80$ | 197 |
| Triglycerides | $144.22 \pm 22.99$ | $205.12 \pm 35.81$ | 143 |
| HDL Cholesterol | $62.49 \pm 14.73$ | $42.32 \pm 8.12$ | 51 |
| LDL Cholesterol | $96.00 \pm 33.58$ | $155.15 \pm 35.83$ | 130 |
| Lipoprotein (a) | $17.33 \pm 5.59$ | $48.16 \pm 15.41$ | 27 |
Abnormalities in Primary Hemostasis: Platelet Dynamics
Platelets are among the first components of the hemostatic system to show alterations in preeclampsia. The disorder is characterized by a “hyper-destructive” state where platelets are consumed at a higher rate than they are produced, leading to a decline in the absolute count and an increase in activation markers.
Consumptive Thrombocytopenia
Thrombocytopenia, defined as a platelet count below $150 \times 10^9/L$, occurs in approximately 15% to 20% of severe preeclampsia cases. The mechanism is primarily consumptive, involving increased platelet utilization at sites of microvascular injury and within the fibrin-rich thrombi that develop in the placental, renal, and hepatic circulations. As platelets are consumed, the bone marrow increases production, releasing younger and larger platelets into the circulation, which explains the observed increase in mean platelet volume (MPV) in preeclamptic patients. In the most severe form, HELLP syndrome, the platelet count can drop precipitously below $50 \times 10^9/L$, presenting an urgent risk for maternal hemorrhage.
Platelet Activation and Surface Markers
Even in preeclamptic women with normal platelet counts, evidence of extensive platelet activation is often present. This is assessed via flow cytometry, which reveals increased expression of surface antigens such as P-selectin (CD62P) and CD63. P-selectin is an adhesion molecule stored in the alpha-granules of platelets; its translocation to the cell surface indicates degranulation. Increased levels of circulating soluble P-selectin and $\beta$-thromboglobulin further confirm this chronic state of activation.
Another important finding is the increased formation of platelet-leukocyte aggregates. Activated platelets bind to monocytes and neutrophils, a process that amplifies the systemic inflammatory response and facilitates the infiltration of leukocytes into vascular tissues, further exacerbating endothelial damage. Longitudinal studies have suggested that an increase in platelet CD63 expression as early as the first trimester can predict the subsequent development of preeclampsia.
Activation of the Coagulation Cascade
In preeclampsia, the systemic coagulation system is in a state of continuous activation, often manifesting as a “low-grade” or “compensated” disseminated intravascular coagulation (DIC). This is demonstrated by the elevation of molecular markers signifying thrombin generation and the consumption of natural inhibitors.
Thrombin Generation and TAT Complexes
Thrombin is the pivotal enzyme of the cascade, responsible for converting fibrinogen to fibrin and activating Factors V, VIII, XI, and XIII. In preeclampsia, markers of in vivo thrombin generation are significantly elevated compared to normal pregnancy. These markers include thrombin-antithrombin (TAT) complexes and prothrombin fragments 1 and 2 ($F_{1+2}$). TAT complexes are formed when antithrombin III neutralizes thrombin; thus, their concentration is a direct reflection of thrombin activity. Studies have shown that TAT levels rise significantly throughout gestation in preeclamptic women, with a notable difference from normotensive controls becoming apparent as the disease progresses.
Consumption of Natural Anticoagulants
The over-activation of coagulation leads to the exhaustion of natural anticoagulants. Antithrombin III (ATIII) is the most critical inhibitor, and its levels are typically reduced in preeclampsia due to the formation of TAT complexes and possibly localized consumption in the placental microvasculature. A decrease in ATIII is often considered a reliable early marker of preeclampsia severity.
Protein C and Protein S also show diminished activity. Protein C is activated by thrombin when bound to thrombomodulin on the endothelial surface; its role is to inactivate Factors Va and VIIIa. In preeclampsia, the reduction in Protein C activity suggests an impaired ability to regulate thrombin generation. Furthermore, there is a significant reduction in free Protein S, the cofactor for Protein C, which is already naturally lower in pregnancy.
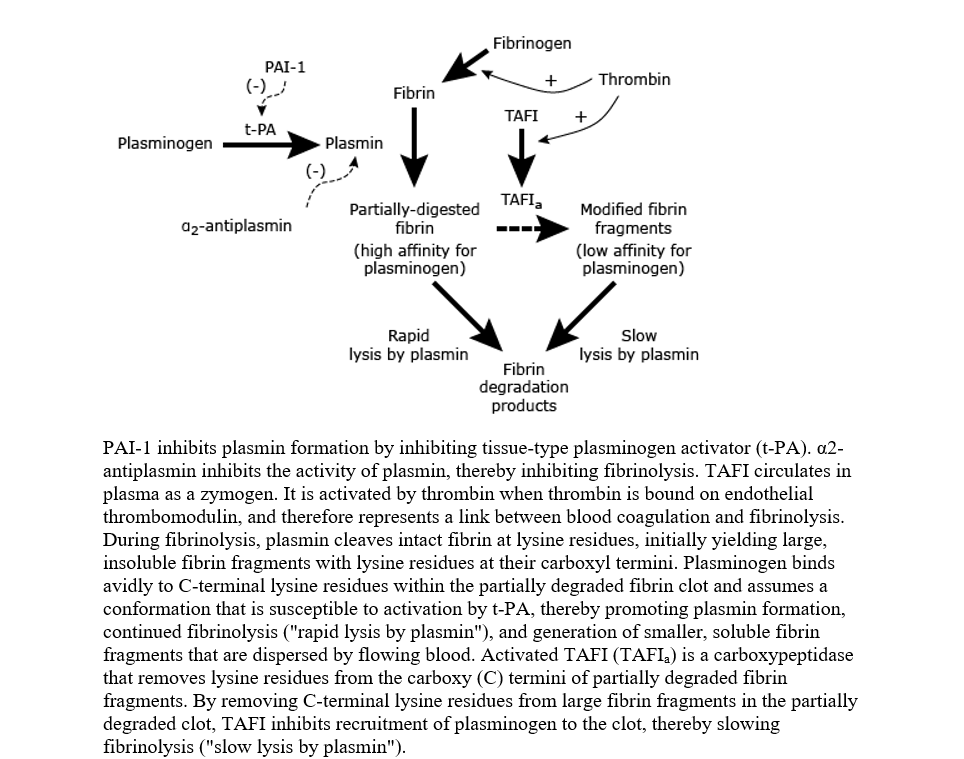
Fibrinolysis and Its Regulation
The fibrinolytic system, responsible for the dissolution of fibrin clots, is highly compromised in preeclampsia. This dysfunction is primarily mediated by an imbalance in the levels of plasminogen activator inhibitors (PAIs) and the thrombin-activatable fibrinolysis inhibitor (TAFI).
The PAI-1 to PAI-2 Ratio as a Clinical Indicator
In a normal pregnancy, both PAI-1 and PAI-2 increase progressively. PAI-1 is synthesized mainly by endothelial cells and platelets, whereas PAI-2 is uniquely derived from the placenta. Preeclampsia is characterized by a significant elevation in PAI-1, reflecting widespread endothelial injury and activation. Concurrently, PAI-2 levels are often significantly lower than those in normotensive pregnancies, indicating placental insufficiency or placental infarction.
The resulting high PAI-1/PAI-2 ratio serves as a sensitive marker for preeclampsia, particularly the early-onset variant. This imbalance suggests a profound suppression of the fibrinolytic system, which prevents the effective clearing of fibrin deposits in the microcirculation and promotes placental ischemia. Some research has even linked the PAI-1 (-675 4G/5G) promoter polymorphism to an increased genetic risk for preeclampsia, as the 4G allele is associated with higher basal PAI-1 production and a more pronounced pro-thrombotic state.
Thrombin-Activatable Fibrinolysis Inhibitor (TAFI)
TAFI acts by removing C-terminal lysine residues from fibrin, which are essential for the binding of plasminogen and tissue-type plasminogen activator (t-PA). By inhibiting this binding, TAFI effectively slows down the rate of clot lysis. In preeclampsia, reports on TAFI levels are contradictory. Some studies have reported a significant decrease in TAFI antigen, possibly due to consumption during localized intravascular coagulation. Conversely, other investigations found higher TAFI antigen levels in preeclamptic women throughout all three trimesters, suggesting it may contribute to the global impairment of fibrinolysis observed in the disease. These discrepancies highlight the heterogeneity of preeclampsia phenotypes and the potential for TAFI to act as a sensitive marker in specific clinical presentations.
| Fibrinolytic Factor | Normal (3rd Trimester) | Preeclampsia (3rd Trimester) | Interpretation |
| PAI-1 Antigen | $\sim 34.8$ ng/mL | $\sim 136.0$ ng/mL | Endothelial activation |
| PAI-2 Antigen | $\sim 269.0$ ng/mL | $\sim 186.0$ ng/mL | Placental insufficiency |
| TAFI Antigen | $\sim 3.0$ ng/mL | $\sim 3.4$ ng/mL | Impaired clot lysis |
| Fibrin D-dimer | Elevated | Markedly Elevated | Increased fibrin turnover |
Endothelial Damage and Vascular Markers
The widespread disturbance of the maternal vascular endothelium is the hallmark of Stage II preeclampsia. This damage is quantified through various plasma markers that are released into the circulation following endothelial injury.
- Von Willebrand Factor (VWF): VWF is synthesized and stored in the Weibel-Palade bodies of endothelial cells. In preeclampsia, VWF levels are significantly raised, often by 20% to 50% more than the already elevated levels of a normal pregnancy. This elevation facilitates increased platelet adhesion to damaged vessel walls.
- Fibronectin: This glycoprotein is a constituent of the subendothelial matrix. Elevated plasma fibronectin levels serve as a highly specific marker of vascular injury in preeclampsia and often correlate with the severity of the disease.
- Soluble Thrombomodulin: Thrombomodulin is an integral membrane protein on endothelial cells that serves as a cofactor for Protein C activation. In preeclampsia, the shedding of thrombomodulin from the cell surface leads to increased plasma levels, which have been shown to predict hypertensive complications with up to 80% accuracy if measured as early as the 24th week of gestation.
- E-selectin and VCAM-1: These adhesion molecules are upregulated on the endothelial surface in response to inflammatory cytokines. Elevated plasma concentrations of soluble E-selectin and vascular cell adhesion molecule-1 (VCAM-1) further attest to the systemic endothelial activation and dysfunction characteristic of preeclampsia.
Differential Diagnosis of Hemostatic Disorders in Pregnancy
Clinical presentation often overlaps between preeclampsia and other serious hemostatic disorders of pregnancy. Distinguishing these conditions is critical, as management—particularly the decision regarding the timing of delivery and the use of plasma exchange—differs fundamentally.
The Logic of French’s Index in Differential Diagnosis
Using a stepwise diagnostic approach, such as that advocated in French’s Index, clinicians must categorize hemostatic abnormalities based on core clinical features and specific laboratory thresholds.
- Gestational Thrombocytopenia: This is the most common cause of low platelets in pregnancy (70%–80%). It is typically an isolated finding occurring in the late second or third trimester, with platelet counts usually above $70-80 \times 10^9/L$ and no maternal history of bleeding or hypertension.
- HELLP Syndrome: A variant of severe preeclampsia characterized by hemolysis (schistocytes on smear, high LDH), elevated liver enzymes, and low platelets. It must be differentiated from rarer thrombotic microangiopathies.
- Thrombotic Thrombocytopenic Purpura (TTP): This condition involves severe ADAMTS13 deficiency ($<10\%$) and can occur at any stage of pregnancy, including the first trimester, whereas preeclampsia and HELLP are rare before 20 weeks. TTP presents with profound thrombocytopenia (often $<30 \times 10^9/L$) and often more severe neurological impairment.
- Atypical Hemolytic Uremic Syndrome (aHUS): Characterized by profound renal failure (creatinine $>2$ mg/dL) and thrombocytopenia. It is driven by complement dysregulation and often requires treatment with complement inhibitors such as eculizumab.
- Acute Fatty Liver of Pregnancy (AFLP): Presents with jaundice, hypoglycemia, and profound coagulopathy with low fibrinogen levels, often occurring in the late third trimester.
| Condition | Typical Onset | Platelet Count (×109/L) | Hypertension | Major Laboratory Feature |
| Gestational Thrombocytopenia | 3rd Trimester | $70 – 150$ | Absent | Isolated low platelets |
| HELLP Syndrome | $>20$ weeks | $<100$ | Present (85%) | High AST/ALT, Schistocytes |
| TTP | Any trimester | $<30$ | Often Absent | ADAMTS13 $<10\%$ |
| aHUS | Any/Postpartum | $20 – 100$ | Often Present | Creatinine $>2$ mg/dL |
| AFLP | 3rd Trimester | $50 – 150$ | Variable | Low Fibrinogen, Hypoglycemia |
Clinical Management and Hemostatic Considerations
The management of preeclampsia-related hemostatic abnormalities involves balancing the prevention of thrombosis with the significant risk of hemorrhage during the peripartum period.
Aspirin Prophylaxis
Low-dose aspirin (81 mg to 150 mg daily) is the primary prophylactic measure for women at high risk for preeclampsia. Aspirin irreversibly inhibits platelet cyclooxygenase, thereby reducing the synthesis of thromboxane $A_2$. This therapy has been shown to reduce the risk of preeclampsia by approximately 15% to 20% if initiated before the 16th week of gestation.
Magnesium Sulfate and Seizure Prophylaxis
Magnesium sulfate is the pharmacological cornerstone for preventing and treating eclamptic seizures. While it has no significant direct effect on the coagulation cascade, it does possess mild antiplatelet and vasodilatory properties. It is important to note that magnesium sulfate is contraindicated within two hours of delivery for mothers with toxemia if there is significant myocardial damage or heart block.
Herb-Drug Interactions in Preeclampsia
The use of herbal supplements among pregnant women is common, but these substances can significantly interact with the hemostatic system and conventional therapies.
- Garlic (Allium sativum): While garlic can help lower blood pressure and improve endothelial function by increasing nitric oxide bioavailability, it also has a potent blood-thinning effect that can potentiate the action of aspirin or heparin, increasing the risk of maternal hemorrhage.
- Turmeric/Curcumin (Curcuma longa): Curcumin has demonstrated anti-inflammatory and endothelial-protective effects in preclinical models of preeclampsia. However, in large doses, it may act as a uterine stimulant, potentially inducing contractions or bleeding, and its use as a concentrated supplement is generally discouraged during pregnancy.
- Ginger (Zingiber officinale): Used primarily for morning sickness, ginger can inhibit platelet aggregation and may interact with anti-clotting medications.
| Herbal Supplement | Potential Preeclampsia Benefit | Hemostatic Interaction Risk |
| Garlic | Regulates BP, increases NO | Potentiates antiplatelet/anticoagulant drugs |
| Turmeric | Reduces oxidative stress | May inhibit platelet function; uterine stimulant |
| Ginger | Improves vascular function | May prolong bleeding time |
| Shatavari | Enhances NO production | No major known interactions with aspirin |
Nuanced Insights and Long-Term Implications
The hemostatic abnormalities in preeclampsia are far more than just “low platelets.” They represent a systemic shift toward a pro-thrombotic and anti-fibrinolytic state, driven by the placental release of antiangiogenic factors and the subsequent widespread endothelial dysfunction.
Second-Order Implications of the TAT and D-dimer Elevation
While TAT complexes and D-dimers are elevated in normal pregnancy, their exponential rise in preeclampsia indicates a pathological breakdown of the hemostatic balance. This suggests that fibrin is being deposited in the microvasculature at a rate that exceeds the body’s already-compromised ability to clear it. This internal accumulation of fibrin meshes is directly responsible for the mechanical fragmentation of red blood cells (hemolysis) and the progressive ischemic damage to vital organs such as the kidneys and liver.
The Predictive Potential of sFlt-1/PlGF Ratio and Soluble Thrombomodulin
The clinical utility of biomarkers such as the sFlt-1/PlGF ratio and soluble thrombomodulin is rapidly advancing. A ratio of sFlt-1/PlGF $\geq 40$ serves as a powerful predictive tool for identifying high-risk women who will develop preeclampsia within the subsequent weeks. This allows for a more proactive approach to maternal surveillance and the timing of delivery. Similarly, early elevations in soluble thrombomodulin levels may identify those at risk for the most severe endothelial damage, necessitating closer monitoring of renal function and platelet counts.
Future Outlook and Cardiovascular Risk
Preeclampsia is now recognized as a significant predictor of future cardiovascular health. Women who have experienced preeclampsia have a higher likelihood of developing chronic hypertension, ischemic heart disease, and stroke later in life. This suggests that the hemostatic stress test of pregnancy unmasks underlying maternal vascular and genetic vulnerabilities. Longitudinal monitoring of these patients postpartum is essential to mitigate long-term cardiovascular risks.
Conclusion
Abnormalities of hemostasis in preeclampsia are deep-seated, systemic manifestations of placental ischemia and generalized endothelial dysfunction. The disorder disrupts the normal physiological adaptations of pregnancy, leading to a profound hypercoagulable state characterized by excessive thrombin generation, consumptive thrombocytopenia, and a critical suppression of fibrinolysis (high PAI-1/low PAI-2). These hemostatic shifts are responsible for the most severe clinical manifestations, including microangiopathic hemolytic anemia, organ failure, and placental abruption. While low-dose aspirin remains a key preventive strategy, the diagnosis and management of preeclampsia require a high degree of clinical vigilance and a nuanced understanding of the differential diagnosis between various thrombotic microangiopathies. The integration of modern biomarkers and a multidisciplinary approach are vital for optimizing outcomes for both mother and child and for managing the long-term cardiovascular implications of this complex disorder.