Description
Comprehensive Clinical Analysis and Differential Diagnosis of Eosinophilia: A Global Medical Perspective
The clinical evaluation of eosinophilia represents one of the most complex diagnostic pathways in modern internal medicine, requiring a synthesis of immunology, hematology, infectious disease, and organ-specific pathology. Eosinophils are specialized, bone-marrow-derived granulocytes that play a dual role as both protective effector cells against multicellular pathogens and as primary drivers of tissue damage in hypersensitivity and clonal disorders. While their physiological presence is typically homeostatic, their pathological expansion—defined as an increase in the absolute eosinophil count (AEC) beyond established norms—necessitates a rigorous and hierarchical diagnostic approach.
Definition and Quantitative Thresholds of Eosinophilic States
Eosinophilia is quantitatively defined by an increase in the absolute number of eosinophils in the peripheral blood. The standard upper limit of normal is generally accepted as $0.5 \times 10^{9}/L$ (500 cells/$\mu$L), although some laboratories utilize a more stringent threshold of $0.4 \times 10^{9}/L$ when excluding individuals with minor atopic tendencies. It is paramount for the clinician to rely on the absolute count rather than the percentage of total white blood cells, as the latter can be misleading in states of leukocytosis or leukopenia.
The categorization of eosinophilic severity is crucial for assessing the risk of imminent organ dysfunction and determining the urgency of the clinical work-up. Current clinical standards categorize peripheral blood eosinophilia into three distinct levels of severity:
| Eosinophilia Severity | Absolute Eosinophil Count (AEC) | Clinical Correlation and Risk Profile |
| Mild Eosinophilia | $0.5 – 1.5 \times 10^{9}/L$ (500-1500/$\mu$L) | Typically associated with common allergies, asthma, or early-stage drug reactions. Lowest risk for immediate organ damage. |
| Moderate Eosinophilia | $1.5 – 5.0 \times 10^{9}/L$ (1500-5000/$\mu$L) | Requires systematic investigation for parasitic infections, vasculitis, or systemic immune disorders. Moderate risk of tissue infiltration. |
| Severe Eosinophilia | $>5.0 \times 10^{9}/L$ ($>5000/\mu$L) | High risk for severe complications such as endomyocardial fibrosis or neurological deficits. Immediate specialist referral is mandatory. |
| Hypereosinophilia (HE) | $\geq 1.5 \times 10^{9}/L$ | Defined as a persistent state recorded on at least two examinations over an interval of $\geq 2$ to $4$ weeks. |
Hypereosinophilic Syndrome (HES) is a clinical designation reserved for cases where hypereosinophilia is accompanied by objective evidence of eosinophil-mediated organ damage (e.g., cardiac, pulmonary, or gastrointestinal dysfunction), provided that no other secondary cause for that damage can be identified. Notably, tissue eosinophilia can occur in the absence of peripheral blood eosinophilia, particularly in organ-restricted diseases like eosinophilic esophagitis (EoE), where diagnosis relies on biopsy findings rather than CBC results.
Global Epidemiology and Socioeconomic Variability
The prevalence and etiology of eosinophilia exhibit a profound geographic and socioeconomic divide. On a global scale, parasitic infestations, particularly helminthic infections, are the leading cause of elevated eosinophil counts. In contrast, within industrialized nations, the epidemiological landscape is dominated by allergic diseases and drug hypersensitivity.
| Etiological Driver | High-Income/Developed Regions | Low-Middle Income/Developing Regions |
| Primary Cause | Allergic/Atopic Disease | Parasitic Infestation |
| Common Pathogens | Strongyloides (immigrants), Toxocara | Ascaris, Schistosoma, Hookworms |
| Allergic Trends | Increasing EoE and Asthma | Lower, but rising with urbanization |
| Clonal Incidence | Relatively rare but well-documented | Often underdiagnosed due to lack of molecular testing |
Statistical trends indicate a rising incidence of eosinophilic esophagitis worldwide, particularly in North America, Western Europe, and Australia. The global pooled incidence of EoE is approximately 5.31 per 100,000 inhabitant-years, with prevalence rates as high as 74.42 per 100,000 in recent years (2017-2022). This increase outpaces the rise in biopsy rates, suggesting a true environmental shift in immune sensitization. In clinical practice settings, the prevalence of eosinophilia can range from 0.1% to nearly 77.5%, depending on the risk factors of the population being screened.
Molecular Pathophysiology of Eosinophil Expansion
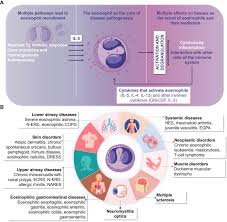
The life cycle of the eosinophil, from myelopoiesis to tissue infiltration and apoptosis, is tightly regulated by a specific triad of cytokines: Interleukin-5 (IL-5), Interleukin-3 (IL-3), and Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF). These cytokines operate through a shared $\beta$-common ($\beta c$) receptor signaling pathway, which is pivotal for understanding both the pathophysiology and the mechanisms of modern targeted therapies.
Cytokine-Mediated Regulation and Hematopoiesis
IL-5 is the most specific and potent cytokine for the eosinophil lineage. While IL-3 and GM-CSF are multi-lineage growth factors involved in the early differentiation of CD34+ pluripotent hematopoietic stem cells into myeloid progenitors, IL-5 is uniquely responsible for the final maturation, selective survival, and activation of eosinophils. The presence of these cytokines prevents programmed cell death (apoptosis) by maintaining the expression of anti-apoptotic proteins, thereby extending the eosinophil’s circulating half-life from hours to several days and facilitating massive tissue accumulation.
Mechanistic Basis of Tissue Destruction
The deleterious effects of eosinophilia are mediated by the release of highly basic, preformed proteins and lipid mediators stored within specialized granules.
The primary cytotoxic mediators include:
- Major Basic Protein (MBP): Potent helminthotoxin and cytotoxin that damages epithelial surfaces and triggers histamine release from mast cells.
- Eosinophil Cationic Protein (ECP): A ribonuclease with potent bactericidal and neurotoxic properties that disrupts cellular membranes.
- Eosinophil Peroxidase (EPX): Catalyzes the formation of hypohalous acids, leading to oxidative stress and tissue necrosis.
- Leukotrienes ($LTC_{4}$): Promote smooth muscle constriction, mucus secretion, and vascular leakage, particularly in the airways.
In sustained hypereosinophilic states, these substances cause irreversible structural damage. For example, in the heart, MBP and ECP lead to subendocardial necrosis followed by thrombus formation and eventually endomyocardial fibrosis. In the lungs, they contribute to the remodeling of the basement membrane and airway hyperresponsiveness.
Hierarchical Classification of Eosinophilic Disorders
The 2024 update of the World Health Organization (WHO) and the International Consensus Classification (ICC) provides a robust framework for distinguishing between reactive and clonal eosinophilias.
- Secondary (Reactive) Eosinophilia
This is the most frequent category, wherein eosinophilia is an appropriate (though potentially damaging) immune response to an external trigger.
- Infections: Primarily helminthic parasites with tissue-migrating phases (e.g., Ascariasis, Trichinellosis, Strongyloidiasis).
- Atopic Diseases: Asthma, allergic rhinitis, and severe eczema.
- Drug Hypersensitivity: Ranging from simple pruritus to life-threatening DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms) syndrome.
- Autoimmune and Vasculitic Disorders: Eosinophilic Granulomatosis with Polyangiitis (EGPA) and systemic lupus erythematosus.
- Paraneoplastic Reactions: Eosinophilia driven by IL-5 production from solid tumors (lung, GI) or Hodgkin’s lymphoma.
- Primary (Clonal) Eosinophilia
This category comprises hematological malignancies where the eosinophil lineage is part of the malignant clone.
- Myeloid/Lymphoid Neoplasms with TK Gene Fusions (MLN-eo-TK): Defined by rearrangements in PDGFRA, PDGFRB, FGFR1, JAK2, or FLT3.
- Chronic Eosinophilic Leukemia (CEL): A myeloproliferative neoplasm involving clonal eosinophilia and bone marrow dysplasia, often excluding the specific fusions seen in MLN-eo-TK.
III. Idiopathic and Specialized Variants
- Idiopathic Hypereosinophilic Syndrome (iHES): A diagnosis of exclusion for persistent HE with organ damage where neither clonal nor reactive causes are found.
- Lymphocytic Variant (L-HES): A reactive state where a clonal T-cell population (e.g., CD3-CD4+) secretes excessive IL-5.
- Hereditary Eosinophilia: A rare, often asymptomatic familial condition.
Clinical Manifestations and Organ System Profiling
The clinical presentation of eosinophilia is multifaceted, ranging from benign incidentalomas to catastrophic multi-organ failure. The specific symptoms are dictated by the target organs of eosinophilic infiltration.
| Organ System | Prevalence in HES | Clinical Presentation and Signs |
| Cutaneous | ~69% | Pruritus, urticaria, angioedema, papulonodular lesions, and mucosal ulcerations (common in myeloid HES). |
| Pulmonary | ~44% | Cough, dyspnea, wheezing, nocturnal asthma, and eosinophilic pleural effusions. |
| Gastrointestinal | ~38% | Dysphagia (EoE), abdominal pain, dyspepsia, weight loss, and eosinophilic colitis. |
| Cardiovascular | ~5-20% | Heart failure, restrictive cardiomyopathy, mural thrombi, and arrhythmias (high mortality risk). |
| Neurological | ~10-15% | Thromboembolic stroke, mononeuritis multiplex, altered mental status, and peripheral neuropathy. |
| Hematologic | >90% | Anemia, thrombocytopenia, and hepatosplenomegaly in clonal forms. |
Cardiovascular Pathology: The Triple-Stage Progression
Cardiac involvement is the most critical complication of hypereosinophilia. It classically progresses from an acute necrotic stage (infiltration and cell death) to a thrombotic stage (large mural thrombi in the ventricles) and finally to a fibrotic stage (restrictive cardiomyopathy). The presence of elevated Troponin, new murmurs, or ECG changes in a patient with eosinophilia should be treated as a medical emergency.
Diagnostic Evaluation and Algorithmic Work-up
The diagnosis of eosinophilia must be systematic, starting with the most common reactive causes before progressing to complex molecular testing.
- Clinical History and Semiological Clues
A thorough history is the single most effective tool. Physicians must prioritize:
- Drug Intake: Allopurinol, carbamazepine, sulfonamides, and penicillin are frequent culprits.
- Travel and Environmental Exposure: History of travel to the tropics or exposure to freshwater; even remote history is relevant for Strongyloidiasis.
- Atopic Context: Personal or family history of asthma, hay fever, or eczema.
- Constitutional Symptoms: Night sweats, fever, and weight loss (“B-symptoms”) are red flags for lymphoma or leukemia.
- Physical Examination Findings
Specific physical findings guide the differential diagnosis:
- Dermatology: Examine for erythema, urticaria, or palpable purpura (suggestive of vasculitis or DRESS).
- Lymphatic System: Hard, fixed nodes suggest malignancy; tender nodes may suggest infection or drug reaction.
- Respiratory: Auscultation for wheeze (asthma, EGPA) or crackles (eosinophilic pneumonia).
- Abdomen: Hepatomegaly or splenomegaly points toward clonal myeloid disorders or systemic mastocytosis.
- Laboratory Investigations
- Blood Film: Essential for identifying blast cells, dysplastic eosinophils, or microfilariae.
- Organ Screening: LFTs (liver involvement), serum creatinine (renal damage), and Troponin (cardiac injury).
- Serology and Microbiology: Stool for ova and parasites ($O\&P$) on three separate occasions; serology for Strongyloides, Schistosoma, and Toxocara.
- Immunology: Total IgE, Vitamin B12, and serum tryptase (elevated in clonal myeloid diseases and mastocytosis).
- Advanced Molecular and Instrumental Testing
If reactive causes are ruled out, clonal markers are sought:
- Genetics: FISH or RT-PCR for FIP1L1-PDGFRA (the most common treatable clonal driver), PDGFRB, and FGFR1.
- Bone Marrow: Histology to assess for $>20\%$ eosinophil infiltration and megakaryocyte atypia.
- Imaging: HRCT of the chest for infiltrates; Echocardiography and Cardiac MRI for endomyocardial fibrosis and thrombi.
- Endoscopy: Biopsy to quantify tissue eosinophils in suspected GI disorders.
Differential Diagnosis: Comparative Analysis
The diagnostic challenge lies in distinguishing between primary clonal disorders and secondary reactive syndromes that mimic them.
| Feature | Allergic/Asthmatic | Helminthic Infection | Clonal Myeloid (CEL) | EGPA (Churg-Strauss) |
| AEC Level | Mild to Moderate | Moderate to Severe | Severe ($>5.0 \times 10^{9}/L$) | Moderate to Severe |
| Organ Damage | Limited to Airways/Skin | Tissue Migration Signs | Multi-organ (Heart/Spleen) | Vasculitic/Neurological |
| B12/Tryptase | Normal | Normal | Elevated | Normal/Elevated |
| Asthma | Essential | Absent | Usually Absent | Diagnostic Criterion |
| Genetic Marker | None | None | PDGFRA/B, FGFR1 | Negative |
| ANCA | Negative | Negative | Negative | Often positive (p-ANCA) |
The “Strongyloides” Warning
It is clinically imperative to exclude Strongyloides stercoralis before starting corticosteroids in any patient with a potential exposure history (including decades-old travel). Corticosteroids can trigger “Hyperinfection Syndrome,” where the parasite rapidly replicates and disseminates, leading to sepsis and multi-organ failure with high mortality.
Therapeutic Management: Pharmacological and Holistic Strategies
The ultimate goal of therapy is the rapid reduction of the AEC to mitigate tissue damage while addressing the specific underlying driver.
- Pharmacotherapeutic Goals
- Immediate Cytoreduction: In severe cases, stabilize AEC to prevent catastrophic degranulation.
- Symptomatic Remission: Resolve rashes, asthma, or abdominal distress.
- Prevention of Fibrosis: Particularly in the heart and lungs.
- Classic Pharmacotherapy and Dosages
- Systemic Corticosteroids
Standard first-line therapy for iHES, L-HES, and reactive syndromes.
- Dose: Prednisone $1$ mg/kg daily (e.g., $60-80$ mg) for initial control, followed by gradual tapering to a maintenance dose of $\approx 10$ mg daily.
- IV Route: Methylprednisolone $1$ g/day for $3$ days in acute cardiac or neurological emergencies.
- Tyrosine Kinase Inhibitors (TKIs)
- Imatinib Mesylate: The cornerstone for FIP1L1-PDGFRA positive patients. It offers rapid remission, often within days.
- Dose: $100$ mg/day (for PDGFRA) to $400$ mg/day (for PDGFRB).
- Targeted Biologics
- Mepolizumab (Anti-IL-5): Approved for steroid-refractory iHES and EGPA.
- Dose: $300$ mg subcutaneously every $4$ weeks.
- Benralizumab (Anti-IL-5R$\alpha$): Targets the receptor directly; effective in eosinophil depletion.
- Second-line Cytotoxic Agents
- Hydroxyurea: Used for steroid-resistant idiopathic HES.
- Dose: $0.5 – 2$ g daily, often combined with low-dose prednisone.
III. Holistic and Non-Pharmacological Interventions
There is a notable lack of clinical evidence supporting herbal or natural supplements for the treatment of eosinophilia.
- Dietary Elimination: For EoE, the Six-Food Elimination Diet (SFED)—removing milk, wheat, eggs, soy, nuts, and seafood—remains the most effective “natural” intervention, though it requires medical oversight and endoscopic follow-up.
- Supplements: Vitamin D and Calcium are often necessary as adjunctive therapy to prevent bone loss in patients on long-term corticosteroids. Iron supplementation may be required if chronic inflammation or restricted diets lead to anemia.
Clinical Warnings: Negative Drug and Supplement Interactions
Patients often utilize vitamins and herbs that can interfere with conventional treatment or exacerbate the underlying condition.
| Substance | Interacting Medication | Clinical Outcome and Mechanism |
| St. John’s Wort | Imatinib | Potent induction of CYP3A4; reduces imatinib levels by $>40\%$, risking treatment failure. |
| Grapefruit Juice | Imatinib/Steroids | Irreversible inhibition of intestinal CYP3A4; leads to significantly increased drug levels and toxicity. |
| Echinacea | Chemotherapy | Induction of CYP3A4/P-gp; may decrease efficacy or increase toxicity depending on the specific agent. |
| Turmeric/Ginger | Anticoagulants | May enhance “blood thinning” effects, increasing bleeding risk in HES patients with thrombi. |
| Corticosteroids | Allopurinol | Increased risk of severe hypersensitivity reactions (DRESS). |
Conclusion and Strategic Recommendations
The differential diagnosis of eosinophilia is a testament to the necessity of precision medicine in modern practice. The transition from identifying a high AEC to achieving a definitive diagnosis requires a multidisciplinary synergy between clinical acumen and molecular diagnostic technologies. Clinicians must prioritize the exclusion of “silent” parasitic infections and the early detection of subclinical cardiac damage, while remaining cognizant of the new genetic classifications that offer life-saving targeted therapies. Through careful history-taking, rigorous laboratory screening, and the judicious application of both biological and pharmacological agents, the potentially devastating consequences of hypereosinophilia can be mitigated, leading to significantly improved patient outcomes.